A single genetic “switch” may be the secret to how the body’s cleanup crew grows up and keeps our organs running smoothly.
Scientists at the University of Liège have identified a crucial genetic regulator that allows macrophages to fully mature and help maintain healthy organs. This regulator, known as MafB, acts as a “molecular switch” that turns specific genes on or off at the right time and in the right cells.
By carefully controlling this genetic activity, MafB enables the development of macrophages that defend the body and support normal organ function. When MafB is missing, macrophages do not work as they should and lose their ability to carry out their protective duties.
Macrophages and Their Shared Immune Identity
Macrophages are vital immune cells present in nearly every tissue in the body. Often described as the body’s ‘cleaning and maintenance team’, they destroy pathogens (biological agents capable of causing disease in a host organism), clear away dead cells and debris, recycle materials such as iron, and help tissues function properly. Although macrophages adjust to the unique needs of different organs, they also retain a shared identity that allows them to perform these core tasks. Until recently, researchers did not fully understand how this common identity is preserved across tissues and even across species.
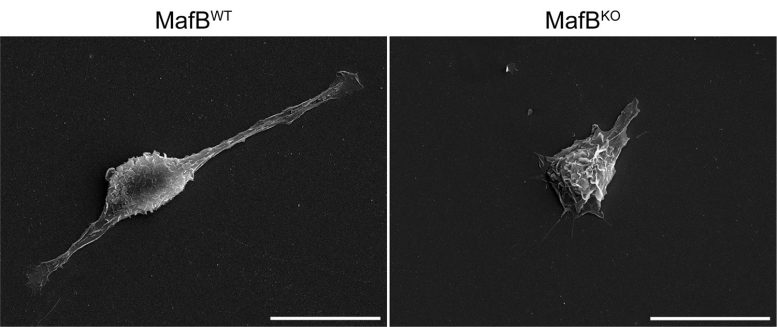
In a study led by Professor Thomas Marichal of the Immunophysiology Laboratory (ULiège), researchers found that MafB serves as a central genetic switch that directs macrophages toward full functionality. As monocytes (immature precursor cells) develop into tissue macrophages, levels of MafB gradually rise, steering the maturation process. Without MafB, these cells remain stuck in an immature state and cannot properly protect the tissues they inhabit.
“Our results show that MafB functions as a master regulator that gives macrophages their identity and equips them with the capabilities necessary to support organ health,” explains immunologist Thomas Marichal. “Without this instruction programme, these cells are present but not fully operational.”
Conserved Genetic Program Across Species
At the molecular level, MafB oversees a broad network of genes essential for macrophage activity. This includes genes involved in phagocytosis (the ability to engulf harmful particles and cellular debris) as well as those responsible for maintaining tissue homeostasis. The researchers found that this genetic program is highly conserved from mice to humans and across vertebrates, underscoring its fundamental role in biology.
The impact of losing this program reaches beyond the immune system itself. When macrophages fail to mature correctly, multiple organs are affected. The study revealed problems with iron recycling in the spleen and disruptions in the normal function of the lungs, intestines, and kidneys. These findings highlight how deeply macrophages influence the body’s overall physiological balance.
“These results reveal that a shared genetic program conserved throughout evolution underlies the specialization of macrophages across tissues,” adds Domien Vanneste, first author of the scientific article. “This explains how these cells can adapt to different organs while preserving their fundamental identity.”
Implications for Chronic Disease Treatment
The discovery also has important medical implications. Malfunctioning macrophages are linked to many chronic conditions, including inflammatory disorders, fibrosis, infections, and metabolic diseases. By targeting MafB or the pathways it regulates, scientists may be able to restore proper macrophage function and improve tissue health in a variety of diseases.
Overall, the findings establish MafB as a central and evolutionarily conserved regulator of macrophage development, identity, and function. The research provides new insight into how the immune system consistently protects and supports multiple organs throughout the body.
Reference: “MafB is a conserved transcriptional regulator of macrophage development and functional identity across tissues and species” by Domien Vanneste, Wen Peng, Zhuangzhuang Liu, Malik Hamaïdia, Raphaël La Rocca, Joan Abinet, Alexis Balthazar, Fabienne Perin, Alexandre Hego, Didier Cataldo, Fabrice Bureau, Philippe Compère, Bénédicte Machiels, Charlotte L. Scott, Coraline Radermecker and Thomas Marichal, 26 February 2026, Immunity.
DOI: 10.1016/j.immuni.2026.01.012
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.