A New Study Discovers a Common Mechanism for Atherosclerosis and Cancer Metastasis
A key molecule for cancer metastasis has been identified as a molecule also involved in cardiovascular disease, suggesting a viable treatment strategy for both diseases at the same time.
Cancer is the uncontrolled development of body cells that results in the creation of tumors, which is caused by the accumulation of mutations in a cell’s DNA. Tumor cells undergo a sequence of alterations that include interactions between the body’s immune system and the tumor in order to become malignant, metastasizing cancer. Many mechanistic elements of this process, however, remain unknown, making cancer prevention and treatment infamously difficult. However, there is accumulating evidence that inflammation of blood vessel-lining “endothelial” cells is a key process in tumor development to metastasis.
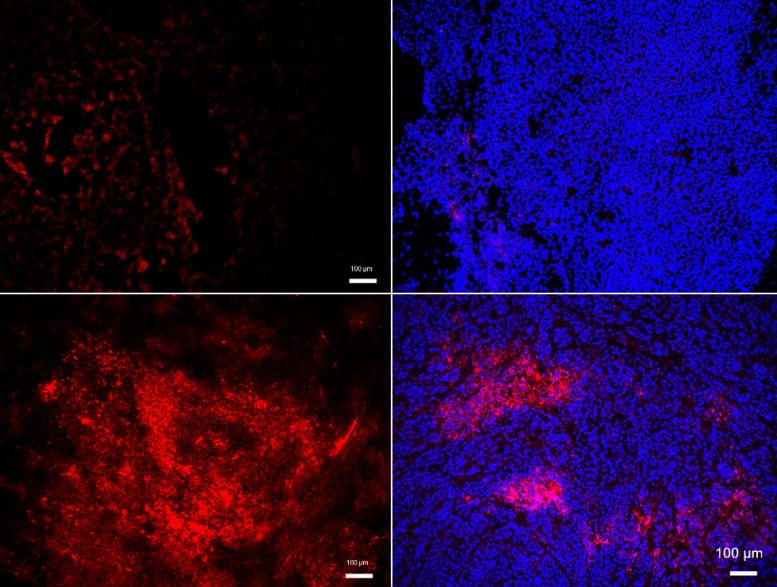
Concerned with the molecular mechanism underlying this process in cancer malignancy, a team of Hokkaido University researchers led by Professor Kyoko Hida discovered that, in malignant tumors, endothelial cells accumulate a lipid delivery molecule known as “low-density lipoprotein” (LDL) and attract immune cells known as “neutrophils.” Neutrophils are immune suppressor cells known to contribute to tumor progression. The finding was recently published in the International Journal of Cancer.
Previous research conducted by the researchers found that blood vessels in malignant tumors exhibited a high number of proteoglycans, and cancerous tissue is known to be inflamed. These characteristics are comparable to those observed in atherosclerosis, and the researchers wanted to explore whether the connections were deeper.
LOX-1 and LDL Accumulation in Metastasizing Tumors
In contrast to non-metastasizing tumors, metastasizing tumors produce proteoglycan molecules, which attach to and accumulate LDL on the walls of blood arteries. The LDL that is bound is oxidized. Its receptor, known as “LOX-1,” is likewise abundant in the blood vessel-lining endothelial cells of metastasizing tumors. They discovered that this leads these cells to create inflammatory signals that attract neutrophils. They subsequently demonstrated that LOX-1 inhibition can greatly decrease tumor malignancy in mice, as well as that LOX-1 overexpression causes an increase in signaling molecules that attract neutrophils.

As the team hypothesized, this sequence of interactions observed in malignant tumors is not novel: it occurs in atherosclerosis, the hardening of blood vessels. “Atherosclerosis and cancer appear to be completely different diseases, but they share several common pathophysiological features in the blood vessels,” says Kyoko Hida.
Even though some questions remain open, especially on the mechanism of how neutrophils contribute to cancer malignancy, this study is the first to explicitly prove the mechanistic commonalities between cardiovascular disease and cancer progression and trace the mechanism involving LDL accumulation and LOX-1 expression in in-vivo tumor tissue. “Our present study focused on the importance of LOX-1 in endothelial cells as a common factor between cancer and atherosclerosis,” Hida explains. “The presence of neutrophils in tumors is a telltale sign of tumor progression.”
The study also points to a promising approach for treating and preventing malignant cancer—and cardiovascular disease—by targeting neutrophil recruitment to endothelial cells. Hida concludes: “The number of patients with cancer who die not of cancer, but of cardiovascular events, is increasing. Targeting the LOX-1/oxidized LDL axis might be a promising strategy for the treatment of the two diseases concomitantly.”
Reference: “The oxidized-LDL/LOX-1 axis in tumor endothelial cells enhances metastasis by recruiting neutrophils and cancer cells” by Takuya Tsumita, Nako Maishi, Dorcas Akuba-Muhyia Annan, Mohammad Alam Towfik, Aya Matsuda, Yasuhito Onodera, Jin-Min Nam, Yasuhiro Hida and Kyoko Hida, 24 May 2022, International Journal of Cancer.
DOI: 10.1002/ijc.34134
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.