
New research clarifies how the brain recognizes familiar stimuli by identifying a brief spike in neural activity (VEP) followed by overall suppression.
Since determining what we observe as new or familiar is essential for prioritizing our attention, neuroscientists have dedicated years to understanding why our brains excel at this task.
During their research, they have encountered seemingly conflicting findings. However, a recent study reveals that these perplexing results are really two sides of the same coin, paving the way for a long-sought understanding of “visual recognition memory” (VRM).
VRM is the ability to quickly recognize the familiar things in scenes, which can then be de-prioritized so that we can focus on the new things that might be more important in a given moment.
Imagine you walk into your home office one evening to respond to an urgent, late email. There you see all the usual furniture and equipment—and a burglar. VRM helps ensure that you’d focus on the burglar, not your bookshelves or your desk lamp.
“Yet we do not yet have a clear picture of how this foundational form of learning is implemented within the mammalian brain,” wrote Picower Professor Mark Bear and fellow authors of the new study in the Journal of Neuroscience.
As far back as 1991 researchers found that when animals viewed something familiar, neurons in the cortex, or the outer layer of their brain, would be less activated than if they saw something new (two of that study’s authors later became Bear’s colleagues at MIT, Picower Professor Earl K. Miller and Doris and Don Berkey Professor Bob Desimone).
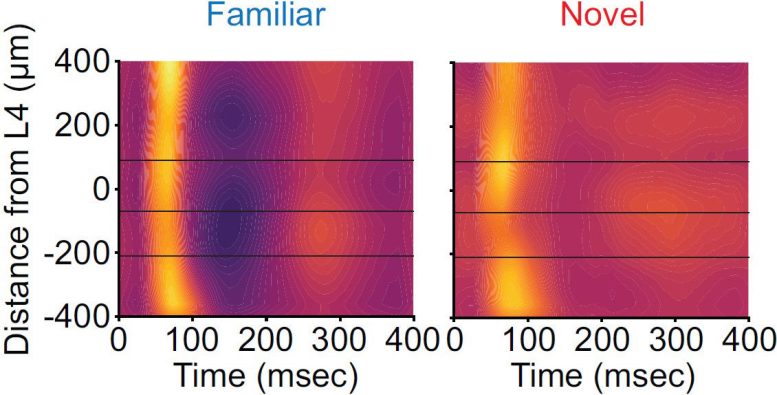
But in 2003, Bear’s lab happened to observe the opposite: Mice would actually show a sharp jump in neural activity in the primary visual region of the cortex when a familiar stimulus was flashed in front of the animal. This spike of activity is called a “visually evoked potential” (VEP), and Bear’s lab has since shown that increases in the VEPs are solid indicators of VRM.
The findings in the new study, led by former Bear Lab postdocs Dustin Hayden and Peter Finnie, explain how VEPs increase even amid an overall decline in neural response to familiar stimuli (as seen by Miller and Desimone), Bear said. They also explain more about the mechanisms underlying VRM – the momentary increase of a VEP may be excitation that recruits inhibition, thereby suppressing activity overall.
New Understanding
Bear’s lab evokes VEPs by showing mice a black-and-white striped grating in which the stripes periodically switch their shade so that the pattern appears to reverse. Over several days as mice view this stimulus pattern, the VEPs increase, a reliable correlate of the mice becoming familiar with—and less interested in—the pattern. For 20 years Bear’s lab has been investigating how the synapses involved in VRM change by studying a phenomenon they’ve dubbed “stimulus-selective response plasticity” (SRP).
Early studies suggested that SRP occurs among excitatory neurons in layer 4 of the visual cortex and specifically might require the molecular activation of their NMDA receptors.
The lab had seen that knocking out the receptors across the visual cortex prevented the increase in VEPs and therefore SRP, but a follow-up in 2019 found that knocking them out just in layer 4 had no effect. So, in the new study, they decided to study VEPs, SRP, and VRM across the whole visual cortex, layer by layer, in search of how it all works.
What they found was that many of the hallmarks of VRM, including VEPs, occur in all layers of the cortex but that it seemed to depend on NMDA receptors on a population of excitatory neurons in layer 6, not layer 4. This is an intriguing finding, the authors said, because those neurons are well connected to the thalamus (a deeper brain region that relays sensory information) and to inhibitory neurons in layer 4, where they had first measured VEPs.
They also measured changes in brain waves in each layer that confirmed a previous finding that when the stimulus pattern is new, the prevailing brain wave oscillations are in a higher “gamma” frequency that depends on one kind of inhibitory neuron, but as it becomes more familiar, the oscillations shift toward a lower “beta” frequency that depends on a different inhibitory population.
A Short Spike Amid a Long Lull
The team’s rigorous and precise electrophysiology recordings of neural electrical activity in the different layers also revealed a potential resolution to the contradiction between VEPs and the measures of labs like that of Miller and Desimone.
“What this paper reveals is that everybody is right,” Bear quipped.
How so? The new data show that VEPs are very pronounced but transient spikes of neural electrical activity that occur amid a broader, overall lull of activity. Previous studies have reflected only the overall decrease because they have not had the temporal resolution to detect the brief spike. Bear’s team, meanwhile, has seen the VEPs for years but didn’t necessarily focus on the surrounding lull.
The new evidence suggests that what’s happening is that VEP is a sign of the activity of the brain quickly recognizing a familiar stimulus and then triggering an inhibition of activity related to it.
“What I think is exciting about this is that it suddenly sheds light on the mechanism, because it’s not that the encoding of familiarity is explained by the depression of excitatory synapses,” Bear said. “Rather, it seems to be accounted for by the potentiation of excitatory synapses on to neurons that then recruit inhibition in the cortex.”
Even as it advances that understanding of how VRM arises, the study still leaves open questions including the exact circuits involved. For instance, the exact contribution of the layer 6 circuit neurons is not yet clear, Bear said. And so, the quest goes on.
Reference: “Electrophysiological signatures of visual recognition memory across all layers of mouse V1” by Dustin J. Hayden, Peter S.B. Finnie, Aurore Thomazeau, Alyssa Y. Li, Samuel F. Cooke and Mark F. Bear, 15 September 2023, JNeurosci.
DOI: 10.1523/JNEUROSCI.0090-23.2023
In addition to Hayden, Finnie, and Bear, the paper’s other authors are Aurore Thomazeau, Alyssa Li, and Samuel Cooke.
The National Eye Institute of the National Institutes of Health, The Picower Institute for Learning and Memory, and The JPB Foundation funded the study.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Boo, stop testing on animals. Your research is moot and you’re a barbarian. The article then becomes garbage, congratulations.