Why do we freeze when startled? New study in flies points to serotonin.
Columbia researchers uncover the mechanism that produces the fly’s startle response, offers clues as to what may happen in our own bodies when we get startled.
A Columbia University study in fruit flies has identified serotonin as a chemical that triggers the body’s startle response, the automatic deer-in-the-headlights reflex that freezes the body momentarily in response to a potential threat. Today’s study reveals that when a fly experiences an unexpected change to its surroundings, such as a sudden vibration, the release of serotonin helps to literally — and temporarily — stop the fly in its tracks.
These findings, published today in Current Biology, offer broad insight into the biology of the startle response, a ubiquitous, yet mysterious, phenomenon that has been observed in virtually every animal studied to date, from flies to fish to people.
“Imagine sitting in your living room with your family and — all of a sudden — the lights go out, or the ground begins to shake,” said Richard Mann, Ph.D., a principal investigator at Columbia’s Mortimer B. Zuckerman Mind Brain Behavior Institute and the paper’s senior author. “Your response, and that of your family, will be the same: You will stop, freeze and then move to safety. With this study, we show in flies that a rapid release of the chemical serotonin in their nervous system drives that initial freeze. And because serotonin also exists in people, these findings shed light on what may be going on when we get startled as well.”
Videos of flies using FlyWalker, a program that enables scientists to label and track the position of each of the fly’s footfalls, thereby building a high-resolution picture of it’s walking gait. Top: normal fly walking at around 25 mm/s. Bottom: fly with its VNC serotonin neurons stimulated, which slows its speed to 15 mm/s. Credit: Clare Howard/Mann lab/Columbia University’s Zuckerman Institute
In the brain, serotonin is most closely associated with regulating mood and emotion. But previous research on flies and vertebrates has shown it can also affect the speed of an animal’s movement. The Columbia researchers’ initial goal was to more fully understand how the chemical accomplished this.
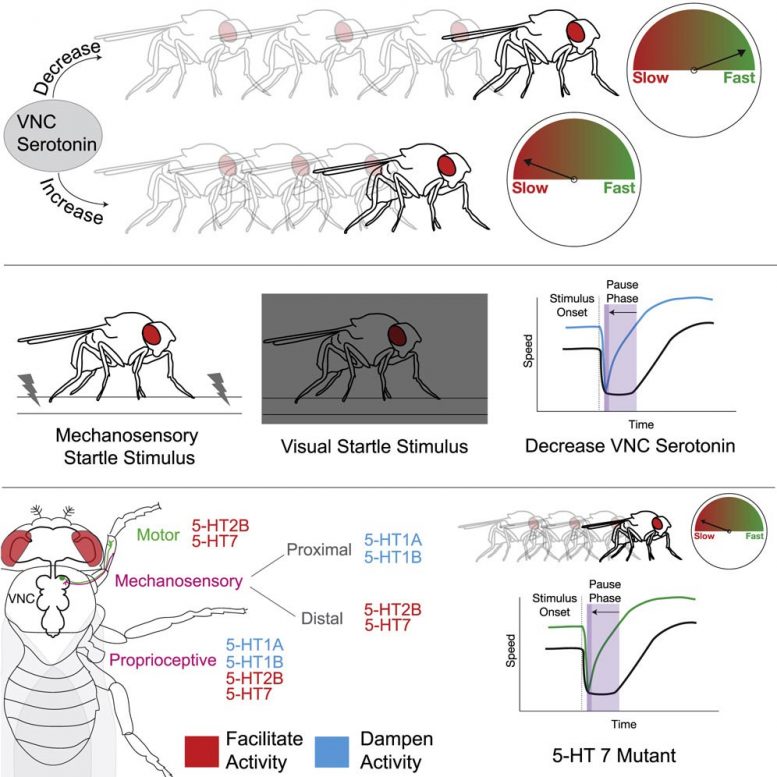
The team first analyzed fruit fly steps using FlyWalker, an apparatus developed by Dr. Mann and Columbia physicist Szabolcs Marka, Ph.D., to track an insect’s steps on a special type of glass. After monitoring how the flies moved, the scientists manipulated the levels of serotonin — and another chemical called dopamine — in the fly’s ventral nerve cord (VNC), which is analogous to the vertebrate spinal cord.
Their initial results revealed that activating neurons that produce serotonin in the VNC slows flies down, while silencing those same neurons speeds flies up. Additional experiments showed that serotonin levels could impact the insects’ walking speed under a wide variety of conditions, including different temperatures, when the flies were hungry, or while they walked upside down, all situations that normally affect walking speed.
“We witnessed serotonin’s biggest effects when the flies experienced rapid environmental changes,” said Clare Howard, Ph.D., the paper’s first author. “In other words, when they were startled.”
To further investigate, the research team devised two scenarios to elicit a fly’s startle response. In the first, they turned the lights off: a total blackout for the insects. For the second, they simulated an earthquake.
To accomplish this, the scientists partnered with Tanya Tabachnik, Director of Advanced Instrumentation at Columbia’s Zuckerman Institute. Tabachnik’s team of machinists and engineers works with scientists to design and build customized systems for their research. For this study, they created a miniature, fly-sized arena perched atop specialized vibrating motors. Adjusting the motors’ strength produced the desired earthquake effect. When the researchers exposed the flies to either the blackout or earthquake scenarios, they also manipulated the fly’s ability to produce serotonin.
“We found that when a fly is startled in these scenarios, serotonin acts like an emergency brake; its release is needed for them to freeze, and that part of this response may be a result of stiffening both sides of the animal’s leg joints,” said Dr. Mann, who is also the Higgins Professor of Biochemistry and Molecular Biophysics (in Systems Biology) at Columbia’s Vagelos College of Physicians and Surgeons. “This co-contraction could cause the brief pause in walking, after which the insect begins to move.”
“We think this pause is important,” added Dr. Howard, “It could allow the fly’s nervous system to gather the information about this sudden change and decide how it should respond.”
Interestingly, even though the fly’s response in both scenarios was to cause an immediate pause, their subsequent walking speeds differed significantly.
“After being startled in the blackout scenario, the fly’s gait was slow and deliberate,” Dr. Howard said. “But the earthquake caused the flies to walk faster after the initial pause.”
While these findings are specific to fruit flies, the ubiquity of serotonin and the startle response provides clues as to the chemical and molecular processes that occur when more complex animals, including people, get startled.
Going forward, the researchers hope to further investigate serotonin’s role in the movement, as well as what other factors may be at play.
“Our results indicate that serotonin has the potential to interact with many different types of nerve cells in the fly nervous system, such as those that guide movement and process sensory information,” said Dr. Mann. “As we and others continue to investigate, we hope to develop a detailed, molecular blueprint for locomotion that can be applied broadly to other animals, perhaps even people.”
Reference: “Serotonergic modulation of walking in Drosophila” by Clare E. Howard, Chin-Lin Chen, Tanya Tabachnik, Rick Hormigo, Pavan Ramdya and Richard S. Mann, 27 November 2019, Current Biology.
DOI: 10.1016/j.cub.2019.10.042
This research was supported by BRAIN Initiative of the National Institutes of Health (1U01NS090514, 1U19NS104655), the Columbia MD/Ph.D. Training Program (GM007367), the Columbia Neuroscience Program (5T32NS065928) and the Swiss National Science Foundation (31003A_175667).
The authors report no financial or other conflicts of interest.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.