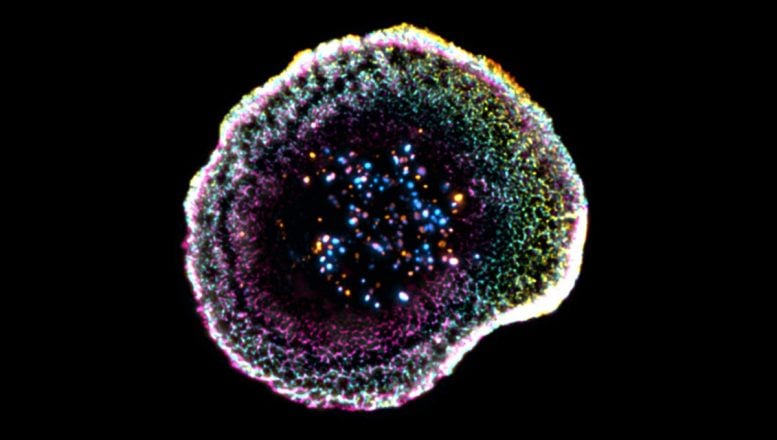
Scientists just uncovered how the immune system’s killer cells strike cancer with microscopic precision.
Cytotoxic T lymphocytes serve as the immune system’s specialized “killer” cells, targeting and eliminating infected or cancerous cells with remarkable accuracy. Their effectiveness depends on a tightly controlled contact point known as the “immune synapse,” where they release toxic molecules that destroy the target while leaving nearby healthy cells unharmed. Until recently, scientists could not clearly observe how these structures are organized.
Researchers at the University of Geneva (UNIGE) and the Lausanne University Hospital (CHUV) have now visualized these processes in three dimensions while preserving conditions close to their natural state. Their findings, published in Cell Reports, show how the internal organization of cytotoxic T cells supports their function and point to new possibilities in immuno-oncology.
How Killer T Cells Precisely Eliminate Threats
When the body encounters infection or cancer, cytotoxic T lymphocytes attach to the affected cell and form the immune synapse. Through this interface, they release toxic compounds that trigger the death of the target cell. This highly controlled process allows the immune system to remove harmful cells without damaging surrounding tissue.
Although the overall mechanism is well known, studying its fine structure at the nanometer scale inside intact human cells has been difficult. One major challenge comes from sample preparation, which can distort delicate cellular features. Traditional imaging methods often require compromises between resolution, the size of the area observed, and the preservation of natural structures.
Cryo-Expansion Microscopy Reveals Hidden Cellular Details
To address these challenges, the UNIGE and CHUV-UNIL team, supported by the ISREC Foundation TANDEM program, used cryo-expansion microscopy (cryo-ExM). “This technique involves instantaneously freezing cells at very high speed, placing them in a so-called vitreous state, where water solidifies without forming crystals and thus faithfully preserves biological structures. The samples are then physically expanded using an absorbent hydrogel, making it possible to observe their internal organization with great precision while maintaining their near-native architecture,” explains Virginie Hamel, Senior Lecturer in the Department of Molecular and Cellular Biology at the Faculty of Science of UNIGE.
With this method, researchers identified new structural features at the point where the immune cell meets its target. “Our work reveals that at the point of contact between the immune cell and its target, the membrane forms a kind of dome, whose structure appears to be linked to adhesion interactions and to the internal organization of the cell,” says Florent Lemaître, postdoctoral researcher in the Department of Molecular and Cellular Biology at the Faculty of Science of UNIGE and first author of the study. The team also examined cytotoxic granules –responsible for destroying target cells – in unprecedented detail. These granules were found to vary in structure, sometimes containing one or multiple “cores” where the active molecules are concentrated.
Imaging Immune Cells Inside Human Tumors
The researchers extended their work beyond isolated cells by applying the technique to human tumor samples. “We extended this approach to human tumor tissues, making it possible to directly observe T lymphocytes infiltrating tumors and their cytotoxic machinery at the nanometer scale. This allows us to study immune responses directly in their clinical context and to better understand the mechanisms that determine their effectiveness,” explains Benita Wolf, Chief Resident and Associate Researcher in the Department of Clinical Oncology at CHUV, who co-led the study.
By offering a three-dimensional view of these processes in conditions close to their natural state, this research provides a valuable framework for understanding how immune cells function. These insights could support the development of improved therapies, especially in immuno-oncology, by clarifying what drives successful immune responses and what limits their effectiveness.
Reference: “Unveiling the molecular architecture of T cells and immune synapses with cryo-expansion microscopy” by Florent Lemaître, Olivier Mercey, Isabelle Mean, Elise Paulin, Valérie Dutoit, Jan A. Rath, Christine von Gunten, Denis Migliorini, Caroline Arber, Paul Guichard, Virginie Hamel and Benita Wolf, 1 April 2026, Cell Reports.
DOI: 10.1016/j.celrep.2026.117165
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.