Researchers have uncovered a fast-acting brain network that may determine how effectively deep brain stimulation improves Parkinson’s symptoms.
Parkinson’s disease can make everyday movements slow, stiff, or shaky, and deep brain stimulation (DBS) is one of the most effective options for easing those motor symptoms. A new study suggests the benefit comes from influencing a specific brain network that communicates most strongly in the fast beta frequency range (20 to 35 Hz). The work was carried out by an interdisciplinary team from the University Hospitals of Cologne and Düsseldorf, Harvard Medical School, and Charité Berlin.
Published in the journal Brain, the research connects two lines of evidence that have often been studied separately. One focuses on electrical activity in the brain, and the other maps where stimulation seems to work best. Bringing these approaches together offers a clearer picture of what DBS may be engaging and why some patients improve more than others.
Linking Space and Time in DBS Research
“For the first time, we were able to characterize the DBS response network in Parkinson’s disease in terms of space and time, simultaneously,” says Professor Dr Andreas Horn from the University of Cologne, who led the study and specializes in computational neurology. “We show that Parkinson’s disease can best be treated if we stimulate a very precisely defined network. This network operates synchronized within a specific frequency band, and offers an explanation for how well patients respond to deep brain stimulation.”
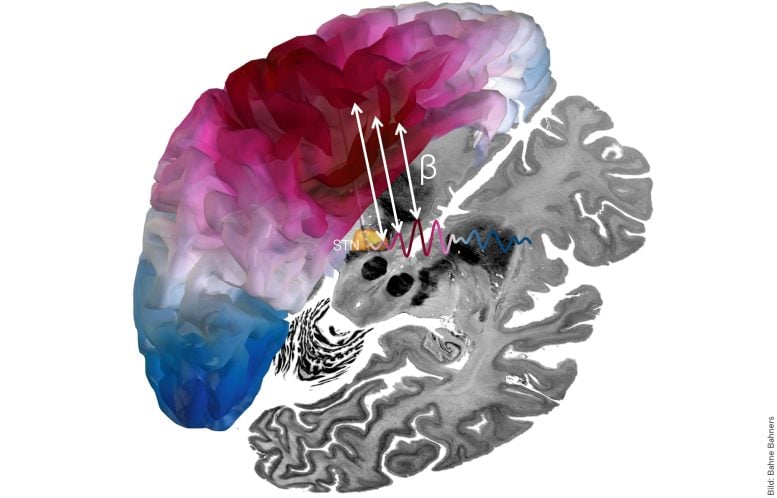
DBS of the subthalamic nucleus is an established way to reduce Parkinson’s motor symptoms by sending small electrical pulses into deep brain tissue. Scientists have long suspected that brain rhythms matter because certain beta patterns are linked to slowed movement in Parkinson’s disease, and DBS can shift those rhythms. Until now, however, studies have tended to capture either the location of the most effective stimulation or the timing and frequency of the signals, without measuring both together in the same framework.
Combining Electrophysiology and Brain Imaging
The research team analyzed data from a large multi-center cohort with one hundred brain hemispheres from fifty patients. Using brain signals that were simultaneously recorded via the implanted DBS electrode and magnetoencephalography (MEG), the scientists mapped the functional connectivity between the deep and superficial areas of the brain.
The study showed that the relevant network between the subthalamic nucleus and frontal brain regions largely communicates at a comparatively fast frequency (20-35 Hz). The strength of this connection explains how well the motor symptoms of individual patients improved after electrode implantation.
“These results suggest that a certain rhythm of the brain acts as a communication channel between the subthalamic nucleus and the cerebral cortex and may mediate the therapeutic effects of deep brain stimulation,” explains Dr Bahne Bahners, first author of the study, who works at Düsseldorf University Hospital. “By stimulating regions that are connected to the identified network, we will probably be able to adjust DBS settings more precisely in the future, especially in patients who have not yet benefited optimally from deep brain stimulation.”
In the future, the researchers plan to take a closer look at the causal effects of deep brain stimulation on brain networks. Studies to this effect are currently being conducted.
Reference: “The deep brain stimulation response network in Parkinson’s disease operates in the high beta band” by Bahne H Bahners, Lukas L Goede, Patricia Zvarova, Garance M Meyer, Konstantin Butenko, Roxanne Lofredi, Nanditha Rajamani, Frederic L W V J Schaper, Clemens Neudorfer, Barbara Hollunder, Julianna Pijar, Savir Madan, Lauren A Hart, Matthias Sure, Alexandra Steina, Fayed Rassoulou, Christian J Hartmann, Markus Butz, Jan Hirschmann, Jan Vesper, Katharina Faust, Gerd-Helge Schneider, Tilmann H Sander, Wolf-Julian Neumann, Michael D Fox, Kai J Miller, Alfons Schnitzler, Andrea A Kühn, Esther Florin and Andreas Horn, 6 February 2026, Brain.
DOI: 10.1093/brain/awaf445
The study was largely funded by the Professor Klaus Thiemann Foundation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.