A groundbreaking brain atlas maps nearly 680,000 cells to reveal how the human brain develops at the single-cell level. The discovery could transform Parkinson’s research by setting new standards for building accurate lab-grown neurons.
Researchers at Duke-NUS Medical School, together with international collaborators, have produced one of the most detailed single-cell maps of the developing human brain to date. This atlas catalogues almost every type of brain cell, documents their genetic signatures, and shows how they grow and communicate with one another. It also evaluates leading laboratory techniques used to generate high-quality neurons, representing a key advance toward new treatments for Parkinson’s disease and other neurological conditions.
Why Parkinson’s disease is a central focus
Parkinson’s disease is the second most common neurodegenerative disorder in Singapore and affects roughly three in every 1,000 people aged 50 and above. The disease destroys midbrain dopaminergic neurons—the cells responsible for releasing dopamine, a chemical essential for movement control and learning. Replacing or restoring these damaged cells could eventually help reduce symptoms such as tremors and loss of mobility.
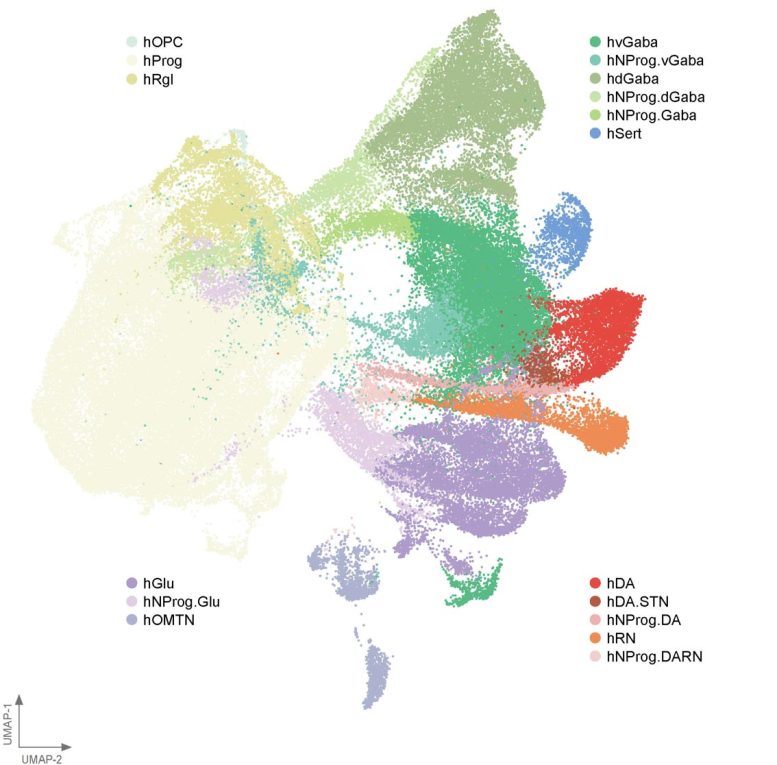
BrainSTEM framework maps hundreds of thousands of brain cells
To better understand how dopaminergic neurons form when grown in the lab, the Duke-NUS team developed a two-step mapping system known as BrainSTEM (Brain Single-cell Two tiEr Mapping). Working with collaborators including the University of Sydney, the researchers examined nearly 680,000 cells from the fetal brain. This large-scale analysis allowed them to map the brain’s full cellular landscape in exceptional detail.
The second stage of the mapping process zooms in on the midbrain and identifies dopaminergic neurons with far greater accuracy. This “comprehensive reference map” gives researchers around the world a reliable benchmark to compare laboratory-grown midbrain models against the real human brain.

Blueprint for improving cell-based therapies
Dr. Hilary Toh, an MD-PhD candidate from the Neuroscience & Behavioural Disorders program at Duke-NUS Medical School and one of the first authors of the study, said: “Our data-driven blueprint helps scientists produce high-yield midbrain dopaminergic neurons that faithfully reflect human biology. Grafts of this quality are pivotal to increasing cell therapy efficacy and minimizing side effects, paving the way to offer alternative therapies to people living with Parkinson’s disease.”
The research, recently published in the journal Science Advances, also revealed a major challenge. Many existing methods for growing midbrain cells were found to unintentionally produce cells from other parts of the brain. These results highlight the need to improve both laboratory protocols and data analysis tools so off-target cells can be identified and removed more effectively.
Enabling AI-driven precision medicine
Dr. John Ouyang, Principal Research Scientist from Duke-NUS’ Centre for Computational Biology and a senior author of the study, said: “By mapping the brain at single-cell resolution, BrainSTEM gives us the precision to distinguish even subtle off-target cell populations. This rich cellular detail provides a critical foundation for AI-driven models that will transform how we group patients and design targeted therapies for neurodegenerative diseases.”
Assistant Professor Alfred Sun from Duke-NUS’ Neuroscience & Behavioural Disorders program, who’s also a senior author of the paper, added: “BrainSTEM marks a significant step forward in brain modeling. By delivering a rigorous, data-driven approach, it will speed the development of reliable cell therapies for Parkinson’s disease. We’re setting a new standard to ensure the next generation of Parkinson’s models truly reflects human biology.”
Open-source tools for global neuroscience research
The team plans to release the brain atlases as open-source resources, along with the multi-tier mapping workflow as a ready-to-use package. Because BrainSTEM can be applied to isolate any cell type in the brain, labs worldwide can use it to gain deeper insights, improve experimental workflows, and accelerate progress across neuroscience research.
Professor Patrick Tan, Senior Vice-Dean for Research at Duke-NUS, said: “This study redefines the benchmark—establishing multi-tier mapping as essential for capturing cellular detail in complex biological systems. By revealing how the human midbrain develops in such detail, we will accelerate Parkinson’s research and cell therapy, delivering better care and offer hope to people living with the disease.”
Reference: “BrainSTEM: A single-cell multiresolution fetal brain atlas reveals transcriptomic fidelity of human midbrain cultures” by Hilary S. Y. Toh, Lisheng Xu, Carissa Chen, Pengyi Yang, Alfred X. Sun and John F. Ouyang, 31 October 2025, Science Advances.
DOI: 10.1126/sciadv.adu7944
The work was supported by funding programs including the USyd-NUS Ignition Grant and the Duke-NUS Parkinson’s Research Fund, made possible through a generous donation from The Ida C. Morris Falk Foundation.
Duke-NUS continues to lead in medical research and education, with a strong focus on advancing patient care through scientific discovery. This study contributes to ongoing efforts to better understand how the human brain works and to develop new therapies for neurological diseases.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.