The Researchers Found That Vitronectin Is a Promising Target for Macular Degeneration
Francesca Marassi, Ph.D., a professor at Sanford Burnham Prebys, is leading research that is shedding light on the molecular mechanisms behind macular degeneration, which accounts for over 90% of all age-related vision loss. The flexible structure of a key blood protein involved in macular degeneration and other age-related disorders, such as Alzheimer’s and atherosclerosis, is described in the research, which was published in the Biophysical Journal.

“Proteins in the blood are under constant and changing pressure because of the different ways blood flows throughout the body,” says Marassi. “For example, blood flows more slowly through small blood vessels in the eyes compared to larger arteries around the heart. Blood proteins need to be able to respond to these changes, and this study gives us fundamental truths about how they adapt to their environment, which is critical to targeting those proteins for future treatments.”
Vitronectin’s Implications for Macular Degeneration
Our blood contains hundreds of proteins, but the researchers concentrated on vitronectin since it is one of the most common. In addition to being present in the blood in large quantities, vitronectin is a crucial component of cholesterol and can be found in the scaffolding between cells.
Vitronectin is involved in numerous age-related disorders, but the most promising target for Marassi’s team is macular degeneration, which affects up to 11 million individuals in the United States. By 2050, this number is projected to double.
“This protein is an important target for macular degeneration because it accumulates in the back of the eye, causing vision loss. Similar deposits appear in the brain in Alzheimer’s disease and in the arteries in atherosclerosis,” says Marassi. “We want to understand why this happens and leverage this knowledge to develop new treatments.”
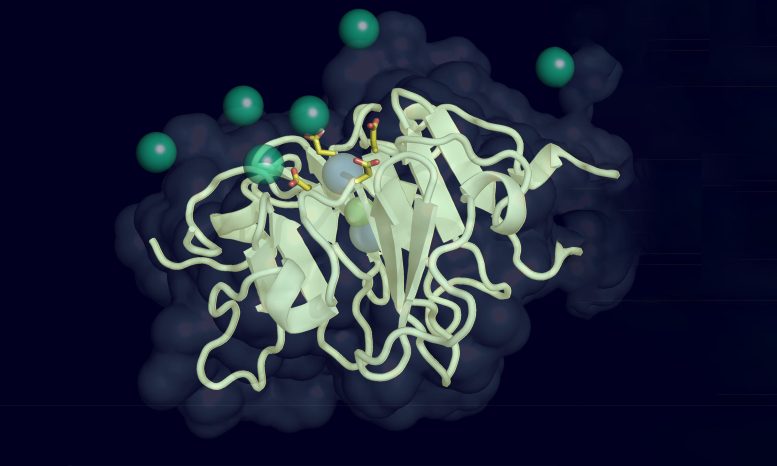
Structural Changes in Vitronectin and Their Consequences
In order to answer this question, the researchers were curious to find out how the protein’s structure altered under various pressures and temperatures, simulating conditions seen in the human body.
“Determining the structure of a protein is the most important part of determining its function,” adds Marassi.
Through detailed biochemical analysis, the researchers found that the protein can subtly change its shape under pressure. These changes cause it to bond more easily to calcium ions in the blood, which the researchers suggest leads to the buildup of calcified plaque deposits characteristic of macular degeneration and other age-related diseases.
“It’s a very subtle rearrangement of the molecular structure, but it has a big impact on how the protein functions,” says Marassi. “The more we learn about the protein on a structural and mechanistic level, the better chance we have of successfully targeting it with treatments.”
These structural insights will streamline the development of treatments for macular degeneration because it will allow researchers and their partners in the biotech industry to custom-design antibodies that selectively block the protein’s calcium-binding without disrupting its other important functions in the body.
“It will take some time to convert it into a clinical treatment, but we hope to have a working antibody as a potential treatment in a few years’ time,” says Marassi. “And since this protein is so abundant in the blood, there may be other exciting applications for this new knowledge that we don’t even know about yet.”
Reference: “Calcium-induced environmental adaptability of the blood protein vitronectin” by Ye Tian, Kyungsoo Shin, Alexander E. Aleshin, Wonpil Im and Francesca M. Marassi, 2 September 2022, Biophysical Journal.
DOI: 10.1016/j.bpj.2022.08.044
The study was funded by the National Institutes of Health, the National Science Foundation, and the Canadian Institutes of Health Research.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
As a senior lay American male victim of some vision loss with some success with at-home nutritional treatments, I can’t dispute that vitronectin could somehow reveal the secrets of so-called “Age Related Macular Degeneration” (AMD). But, why bother researching what is already obvious? Food allergy related acidic blood (e.g. a high serum level of uric acid and a likely calcium deficiency; “Allergy Related Macular Degeneration;” FAMD?) can damage the macula before other areas of the body and brain because blood vessels are densest in the eyes and the eyes are so active during one’s waking period. I imagine it would help if mainstream medicine recognized and researched my kind of allergies and/or could learn from an “experience-based” lay source (as opposed to fatally flawed “evidence-based” professional dogma).