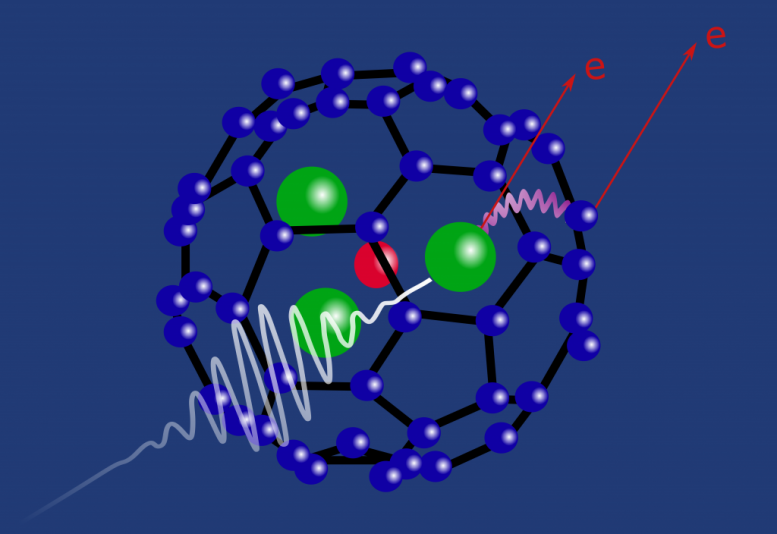
Radiation damage spreads among close neighbors: A single soft x-ray can destroy a protein-sized molecule.
A single x-ray can unravel an enormous molecule, physicists report in the March 17 issue of Physical Review Letters. Their findings could lead to safer medical imaging and a more nuanced understanding of the electronics of heavy metals.
Medical imaging techniques such as MRIs use heavy metals from the bottom of the periodic table as “dyes” to make certain tissues easier to see. But these metals, called lanthanides, are toxic. To protect the person getting the MRI, some chemists wrap the lanthanide inside a cage of carbon atoms.
Molecular physicist Razib Obaid and his mentor, Prof. Norah Berrah in the physics department, wanted to know more about how the lanthanides interact with the carbon cages they’re wrapped in. The cages, 80 carbon atoms strong, are called fullerenes and are shaped like soccer balls. They don’t actually bond to the lanthanide; the metal floats inside the cage. There are many similar situations in nature. Proteins, for example, often have a metal hanging out close to a giant organic (that is, mostly made of carbon) molecule.
“You can induce radiation damage just by striking one atom out of 84.” Razib Obaid
So Obaid and his team of collaborators from Kansas State University, Pulse Institute at Stanford, Max Planck Institute at Heidelberg, and the University of Heidelberg studied how three atoms of the lanthanide element holmium inside of an 80-carbon fullerene reacted to x-rays. Their initial guess was that when an x-ray first hit one of the holmium atoms, it would get absorbed by an electron. But that electron would be so energized by the absorbed x-ray that it would fly right out of the atom, leaving a vacant spot. That spot would then get taken by another of the holmium’s electrons, which would have to jump down from the outer edge of the atom to fill it. That electron had formerly been partnered with another electron on the outskirts of the atom. When it jumped down, its lonely ex, called an Auger electron, would zoom away from the whole molecule and get detected by the scientists. Its distinctive energy would give it away.
It sounds complicated, but that would have been the simplest (and thus most likely) scenario, the physicists thought. But it’s not what they saw.
When Obaid and his colleagues zapped the holmium-fullerene molecule with a soft x-ray (about 160 electron-volts), the number of Auger electrons detected was too low. And too many of the electrons had energies much less than the Auger electrons should have.
After some calculating, the team figured out there was more going on than they’d guessed.
First, the x-ray would hit the holmium, which would lose an electron. The vacant spot would then be filled by the outer edge electron from the holmium atom. That much was correct. But the energy released by the jumping electron (when it jumps ‘down’ from the outskirts of the atom to the interior, it also jumps ‘down’ in energy) would then be absorbed by the carbon fullerene cage or another of the neighboring holmium atoms. In either case, the energy would cause an additional electron to zoom away from whatever absorbed it, the fullerene cage or the holmium atom.
Losing these multiple electrons destabilized the whole molecule, which would then fall apart entirely.
The end result?
“You can induce radiation damage just by striking one atom out of 84,” says Obaid. That is, a single x-ray strike is enough to destroy the entire molecule complex through this energy transfer process involving neighboring atoms. It gives some insight into how radiation damage occurs in living systems, Obaid says. It was always thought that radiation damaged tissue by stripping away electrons directly. This experiment shows that interactions between an ionized atom or molecule and its neighbors can cause even more damage and decay than the original irradiation.
The work also gives medical physicists an idea of how to limit patients’ exposure to heavy metals used as dyes in medical imaging. Shielding all parts of the body from the radiation except for those to be imaged with heavy metal dyes can potentially restrict the heavy metal exposure as well as the radiation damage, the researchers say. The next step of this work would be to understand exactly how fast this interaction with the neighbors occurs. The researchers expect it to take place in just a few femtoseconds (10-15 s).
Reference: “Intermolecular Coulombic Decay in Endohedral Fullerene at the 4d→4f Resonance” by Razib Obaid, Hui Xiong, Sven Augustin, Kirsten Schnorr, Utuq Ablikim, Andrea Battistoni, Thomas J. A. Wolf, René C. Bilodeau, Timur Osipov, Kirill Gokhberg, Daniel Rolles, Aaron C. LaForge and Nora Berrah, 17 March 2020, Physical Review Letters.
DOI: 10.1103/PhysRevLett.124.113002
The work was funded by Department of Energy, Basic Energy Sciences (BES), Division of Chemical Sciences, Geosciences, and Biosciences, under Grant No. DE-SC0012376.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.