Findings may aid design of targeted drug delivery into the brain and central nervous system.
Researchers have developed a zebrafish model to study omega-3 fatty acid transport into the brain, offering insights into lipid transport across the blood-brain barrier and potential drug delivery optimization.
Researchers at the University of California, Los Angeles (UCLA) David Geffen School of Medicine, the Howard Hughes Medical Institute at UCLA, and the National Institutes of Health (NIH) have developed a zebrafish model that provides new insight into how the brain acquires essential omega-3 fatty acids, including docosahexaenoic acid (DHA) and linolenic acid (ALA).
Their findings, published on May 8 in Nature Communications, have the potential to improve understanding of lipid transport across the blood-brain barrier and of disruptions in this process that can lead to birth defects or neurological conditions. The model may also enable researchers to design drug molecules that are capable of directly reaching the brain.
Omega-3 fatty acids are considered essential because the body cannot make them and must obtain them through foods, such as fish, nuts, and seeds. DHA levels are especially high in the brain and important for a healthy nervous system. Infants obtain DHA from breastmilk or formula, and deficiencies of this fatty acid have been linked to problems with learning and memory.
The Blood-Brain Barrier and Lipid Transport
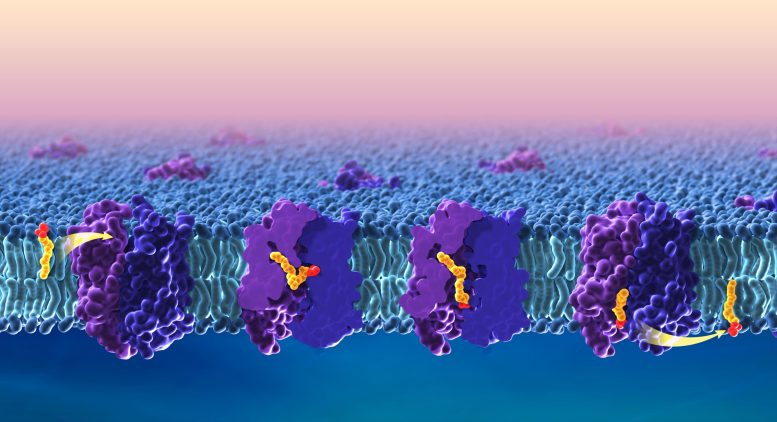
To get to the brain, omega-3 fatty acids must pass through the blood-brain barrier via the lipid transporter Mfsd2a, which is essential for normal brain development. Despite its importance, scientists did not know precisely how Mfsd2a transports DHA and other omega-3 fatty acids.
In the study, the research team provides images of the structure of zebrafish Mfsd2a, which is similar to its human counterpart. The snapshots are the first to detail precisely how fatty acids move across the cell membrane. The study team also identified three compartments in Mfsd2a that suggest distinct steps required to move and flip fatty acids through the transporter, as opposed to movement through a linear tunnel or along the surface of the protein complex.
The findings provide key information on how Mfsd2a transports omega-3 fatty acids into the brain and may enable researchers to optimize drug delivery via this route. The study also provides foundational knowledge on how other members of this transporter family, called the major facilitator superfamily (MFS), regulate important cellular functions.
Reference: “Lipid flipping in the omega-3 fatty-acid transporter” by Chi Nguyen, Hsiang-Ting Lei, Louis Tung Faat Lai, Marc J Gallenito, Xuelang Mu, Doreen Matthies and Tamir Gonen, 8 May 2023, Nature Communications.
DOI: 10.1038/s41467-023-37702-7
The study was led by Tamir Gonen, Ph.D., of UCLA and Doreen Matthies, Ph.D., of NIH’s Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD). Additional funding for the study was provided by NIH’s National Institute of General Medical Sciences (NIGMS) and the Howard Hughes Medical Institute.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.