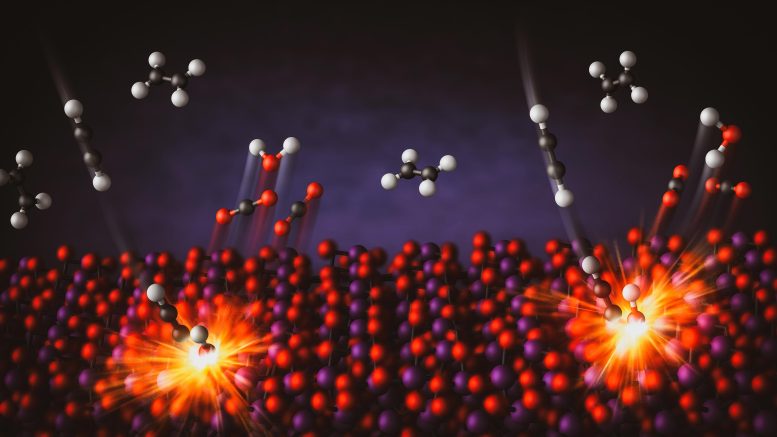
Credit: Greg Stewart/SLAC National Accelerator Laboratory
Researchers have developed a catalyst-driven method to selectively burn one molecule in a hydrocarbon mixture, offering a more efficient way to remove pollutants.
This approach could improve processes in fuel, plastic, and fertilizer production while reducing energy waste.
New Catalyst Discovery for Selective Combustion
For the first time, researchers at the University of Minnesota Twin Cities have developed a new method to selectively burn a specific molecule in a mixture of hydrocarbons — compounds made of hydrogen and carbon.
This breakthrough could make industrial processes more efficient and help remove pollutants. It has potential applications in industries such as fuel production, pharmaceuticals, fertilizers, and plastics.
The research was published on February 13 in Science, a premier multidisciplinary, international peer-reviewed scientific journal.
How the Bismuth Oxide Catalyst Works
The study demonstrates how a bismuth oxide catalyst—a substance that speeds up chemical reactions—can be used to target and burn just one type of molecule in a mixture. The researchers successfully combusted small amounts of acetylene in the presence of ethylene, a key step in preventing catalyst poisoning during polyethylene production. This is particularly important for the plastics industry, which produces over 120 million metric tons of polyethylene each year.
“No one else has shown that you could combust one hydrocarbon present in low concentrations, in mixtures with others,” said Aditya Bhan, a Distinguished McKnight University Professor in the Department of Chemical Engineering and Materials Science and lead investigator on the paper.
Chemical Looping: A Game-Changer
Conventionally, combustion processes are used to burn all hydrocarbon fuel mixtures at high temperatures to produce heat. The use of a catalyst allowed the researchers to tackle the challenge of burning one molecule but not the others. The bismuth oxide catalyst is unique as it provides its own oxygen during combustion, rather than using oxygen from an outside source, in a process called chemical looping.
“We were able to take oxygen out of the catalyst and put it back in multiple times, where the catalyst changes slightly, but its reactivity is not impacted. Operating in this chemical looping mode avoids flammability concerns,” said Matthew Jacob, a University of Minnesota chemical engineering Ph.D. candidate and first author on the paper.
Broader Implications for Industry
Traditionally, eliminating small concentrations of contaminants is very challenging and energy-intensive, but this new method could provide a more energy-efficient alternative.
“You want to do this process selectively. Removing acetylene and other trace hydrocarbon contaminants in this manner could be more energy efficient,” said Matthew Neurock, a professor in the Department of Chemical Engineering and Materials Science and senior co-author on the paper. “You just want to be able to go into a gas mixture to remove some molecules without touching the rest of the molecules.”
A Future of Smarter Catalysis
The researchers said the long-term impact could be high because catalysts are used in the production of just about anything we touch in modern society from fuels and medications to fertilizers and plastics. Understanding how molecules combust—and don’t combust—on catalyst surfaces is valuable for making fuels and plastics production more efficient.
“If we can understand how a catalyst works, at a molecular atomic level, we can adapt it to any particular reaction,” said Simon Bare, a Distinguished Scientist at the SLAC National Accelerator Laboratory at Stanford University, and co-author of the study. “This can help us understand how catalysts, that produce fuels and chemicals needed in modern living, react to their environment.”
Reference: “Selective chemical looping combustion of acetylene in ethylene-rich streams” by Matthew Jacob, Huy Nguyen, Rishi Raj, Javier Garcia-Barriocanal, Jiyun Hong, Jorge E. Perez-Aguilar, Adam S. Hoffman, K. Andre Mkhoyan, Simon R. Bare, Matthew Neurock and Aditya Bhan, 13 February 2025, Science.
DOI: 10.1126/science.ads3181
In addition to Bhan, Jacob, Neurock, and Bare, the University of Minnesota Department of Chemical Engineering and Materials Science team included graduate students Rishi Raj and Huy Nguyen and Professor Andre Mkhoyan, along with Javier Garcia-Barriocanal from the University of Minnesota Characterization Facility. Additional team members included Jiyun Hong, Jorge E. Perez-Aguilar, and Adam S. Hoffman from the SLAC National Accelerator Laboratory at Stanford University.
This work was funded by the U.S. Department of Energy, Office of Basic Energy Sciences. The work was completed in collaboration with the University of Minnesota Characterization Facility and the Minnesota Supercomputing Institute.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.