Two bacteria working in harmony show powerful antitumor effects. The approach could transform treatment for immunocompromised patients.
A research team led by Professor Eijiro Miyako at the Japan Advanced Institute of Science and Technology (JAIST), working in collaboration with Daiichi Sankyo Co., Ltd. and the University of Tsukuba, has created a pioneering bacterial therapy for cancer that operates independently of the immune system. This new approach relies on a specially designed microbial consortium called AUN.
The concept of bacterial cancer therapy dates back to 1868, when German physician Busch reported that intentionally infecting a patient with bacteria led to tumor regression. Later, in 1893, Dr. William Coley proposed bacterial injections as a treatment, laying the foundation for cancer immunotherapy. Over the past 150 years, these early ideas evolved into advanced treatments such as checkpoint inhibitors and CAR-T cells. While effective for some, these therapies all depend on immune cell activity, which limits their effectiveness in patients whose immune systems are weakened by chemotherapy or radiotherapy.

The new AUN therapy overcomes this challenge by combining two naturally occurring bacterial strains:
- Proteus mirabilis (A-gyo), a microbe that thrives within tumors
- Rhodopseudomonas palustris (UN-gyo), a photosynthetic bacterium
Mechanisms of tumor eradication
Working in close cooperation, the “AUN” bacterial pair achieved remarkable tumor clearance in both mouse and human cancer models, even under immunocompromised conditions—all without relying on immune cells. The therapy demonstrated strong biocompatibility and very limited side effects, notably preventing cytokine release syndrome (CRS).

In this study, AUN’s potent antitumor activity was driven by several coordinated bacterial mechanisms, including:
- Precise targeting and destruction of tumor vasculature and cancer cells
- Structural transformation of A-gyo (filamentation) induced by tumor metabolites, which increased its cancer-fighting capacity
- Dynamic intratumoral population shifts, where the initial bacterial ratio of A-gyo : UN-gyo ≈ 3:97 rapidly changed to 99:1 within the tumor microenvironment
- Reduced pathogenicity and minimized side effects, including effective prevention of CRS
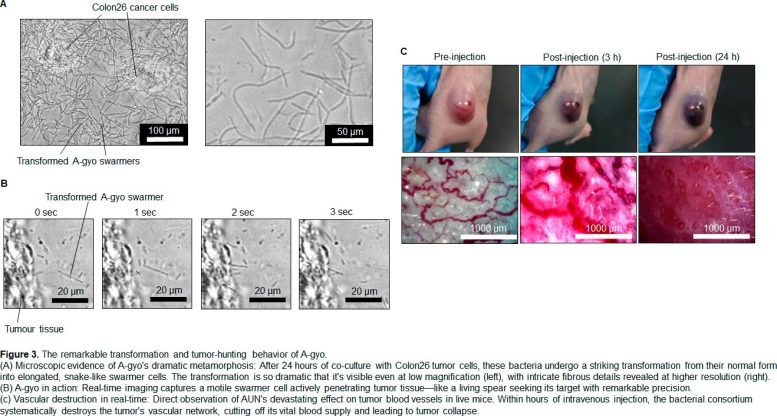
The remarkable transformation and tumor-hunting behavior of A-gyo. Credit: Eijiro Miyako from JAIST
Harmony between bacterial partners
Notably, UN-gyo functions as a regulatory partner only when coexisting with A-gyo, helping to suppress the pathogenicity of both strains while simultaneously enhancing their tumor-specific cytotoxicity. This “cooperation of labor” mirrors the Japanese philosophical concept of “AUN”—perfect harmony between opposites. It is this delicate and dynamic interplay between the two bacterial species that unlocks the remarkable antitumor efficacy—a feat previously unattainable through conventional therapies.
“To accelerate the social implementation of this research, we are preparing to launch a startup and aim to begin clinical trials within six years,” said Professor Eijiro Miyako, lead author of the study.
“A new chapter in bacteria-based cancer therapy—pursued for over 150 years—is finally beginning.”
This revolutionary approach represents a paradigm shift for immunocompromised cancer patients. It offers a long-awaited therapeutic solution in cases where conventional immunotherapies fail—ushering in the dawn of truly immune-independent cancer treatment.
Reference: “Tumour-resident oncolytic bacteria trigger potent anticancer effects through selective intratumoural thrombosis and necrosis” by Seigo Iwata, Taisei Nishiyama, Matomo Sakari, Yuki Doi, Naoki Takaya, Yusuke Ogitani, Hiroshi Nagano, Keisuke Fukuchi and Eijiro Miyako, 5 August 2025, Nature Biomedical Engineering.
DOI: 10.1038/s41551-025-01459-9
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
30 Comments
Your work with cancerous tumors is outstanding, thanks
This is ground breaking news for cancer patients worldwide! There is renewed hope that cancer can be eradicated forever!
Great!!
Great news I new it could be done sooner or later what will big Pharma think about that?
Thank you so much for this information. The information certainly offers HOPE and ENCOURAGEMENT to so many people battling the awful diagnosis of cancer. I am a fan of the work that you do. I will share this information when appropriate with others who might not be aware of these latest advancements/breakthroughs in science. Please keep up the good work.
DITTo
So impressed with your work. Please keep going and hopefully provide hope for the many people suffering with cancer.
Love these NEW more successful and more NATURAL TREATMENTS, without all the Harsh Chemicals & Side Effects. May GOD continue to BLESS and guide all of you who are doing such fantastic work!
Thank you all.
How close is the study to application?
It’ll take within SIX YEARS to even begin clinical trials. People, you might want to look into the two bacteria they are claiming does the trick. Just what IF someone out there much smarter than I didn’t have to rely on these “experts” and “scientists”? And why are you, David, one of the only people here who even thought to ask that intelligent question? Scary how people give up their power without a question or a thought.
Why is it not starting next year y not now people that has it doesn’t have that long
Why is it not starting next year y not now people that has it doesn’t have that long
Love these NEW more successful and more NATURAL TREATMENTS, without all the Harsh Chemicals & Side Effects. May GOD continue to BLESS and guide all of you who are doing such fantastic work!
Thank you all.
can you accept cancer patients who would like for you to try this on them. I have a 61 year old neighbor who has gone through over 1.5 years of chemotherapy. I drive him for labs and this treatment every three weeks. But the prognosis is poor even though the tumor in one Ling and tumor in his liver did shrink over time. He is getting depressed. He needs to try some else to save him please. He prefers text first email second. 989-464-1219
Thankyou for all your hard work and unwavering dedication to helping cancer sufferers world wide
Thank God for everyone of you that are actively searching for safer and positive cancer cures. Your work is so important with your efforts to save lives. God bless you and keep up the good work
God bless and advance your work. Only wish it could be expedited. Patients need groundbreaking treatments, this kind that comes out of smart, motivated start-ups.
Patients need these more groundbreaking treatments coming out of smart, motivated startups such as yours. God bless and expedite your work.
Thank you for the years of study and the hard work you put into this creative option for many.
Would love my brother to be part of a trial as been told no treatment available
Please, please speed up the clinical trial from 6 to 3 years if possible. I cried when I read this article. There is hope. Thank you to the research team.
We hope that treatment to cancer with a painful chemotherapy (causing notable side effects) may be phased out in the near future by the oncologists across the world by adopting this bacteria -based cancer-therapy.
Is there a possibility for bacteria A-Gyo and bacteria UN – Gyo merge or inter react or interbreed at certain conditions evolving a AUN -hybrid bacteria to enhance the efficacy of cancer treatment in-lieu of working as a pair of A-Gyo and UN- Gyo?.
Pray for your continued success. Thank you for giving HOPE.
It’s nice but … “To accelerate the social implementation of this research, we are preparing to launch a startup and aim to begin clinical trials within SIX YEARS,” said Professor Eijiro Miyako, lead author of the study.
I’ve heard that molecular hydrogen water tablets also combat c*ncer – take that information and do your own research.
“The new AUN therapy overcomes this challenge by combining two naturally occurring bacterial strains:
Proteus mirabilis (A-gyo), a microbe that thrives within tumors
Rhodopseudomonas palustris (UN-gyo), a photosynthetic bacterium”
Since clinical trials will take “within” SIX YEARS to begin, I think there has got to be a way to get these “naturally occurring bacterial strains” into the body. Is there some law against that? Or will people go to jail if they take matters out of the hands of these “scientists” and “experts” and “researchers” to find the answer themselves?
Found this on the one bacteria:
Rhodopseudomonas palustris is a rod-shaped, Gram-negative purple nonsulfur bacterium, notable for its ability to switch between four different modes of metabolism.
Rhodopseudomonas palustris is found extensively in nature, and has been isolated from swine waste lagoons, earthworm droppings, marine coastal sediments, and pond water. Although purple nonsulfur bacteria are normally photoheterotrophic, R. palustris can flexibly switch among any of the four modes of metabolism that support life: photoautotrophic, photoheterotrophic, chemoautotrophic, or chemoheterotrophic.
Found this on the other bacteria:
“Proteus mirabilis primarily comes from soil, sewage, and the intestinal flora of humans and many other mammals. It is commonly found in human and animal intestines and is frequently present in soil and wastewater, where it helps decompose organic matter.”
Note: This bacteria is found in urinary tract infections, I’ve read. However, correlation is not indicative of causation. If you find a fireman at the burning house, you don’t say – Oh, yes, the fireman caused the fire.
I pray they are right, but who knows…. What I want to know is, how the researchers discovered this… What made them ever come up with the idea of combining these two bacteria in the first place?
Why is it not starting next year y not now people that has it doesn’t have that long
As long as “they” can make money from the treatment, it will be allowed………
There have been cures to cancers and autoimmune diseases for decades…