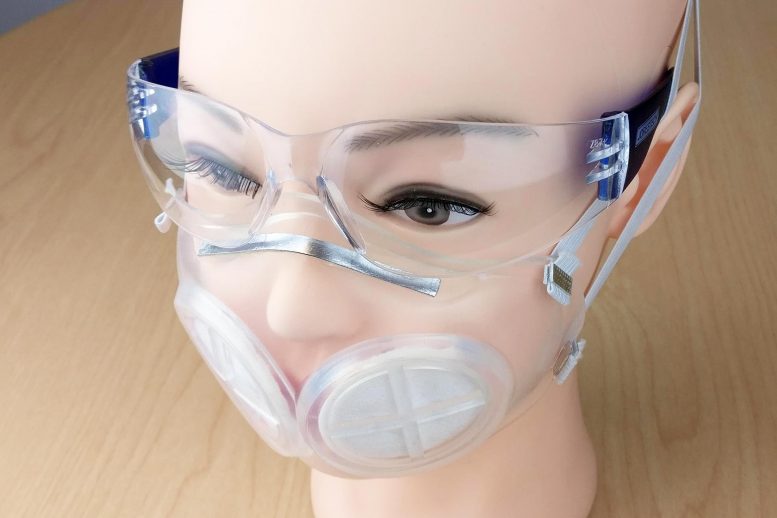
The prototype mask, which includes an N95 filter, can be easily sterilized and worn many times.
Researchers at MIT and Brigham and Women’s Hospital have designed a new face mask that they believe could stop viral particles as effectively as N95 masks. Unlike N95 masks, the new masks were designed to be easily sterilized and used many times.
As the number of new COVID-19 cases in the United States continues to rise, there is still an urgent need for N95 masks for health care workers and others. The new mask is made of durable silicone rubber and can be manufactured using injection molding, which is widely used in factories around the world. The mask also includes an N95 filter, but it requires much less N95 material than a traditional N95 mask.
“One of the key things we recognized early on was that in order to help meet the demand, we needed to really restrict ourselves to methods that could scale,” says Giovanni Traverso, an MIT assistant professor of mechanical engineering and a gastroenterologist at Brigham and Women’s Hospital. “We also wanted to maximize the reusability of the system, and we wanted systems that could be sterilized in many different ways.”
The team is now working on a second version of the mask, based on feedback from health care workers, and is working to establish a company to support scaled-up production and seek approval from the FDA and the National Institute for Occupational Safety and Health (NIOSH).
Traverso is the senior author of a paper describing the new masks, which was published on July 7, 2020, in the British Medical Journal Open. The lead authors of the study are James Byrne, a radiation oncologist at Brigham and Women’s Hospital and research affiliate at MIT’s Koch Institute for Integrative Cancer Research; Adam Wentworth, a research engineer at Brigham and Women’s Hospital and a research affiliate at the Koch Institute; Peter Chai, an emergency medicine physician at Brigham and Women’s Hospital; and Hen-Wei Huang, a research fellow at Brigham and Women’s Hospital and a postdoc at the Koch Institute.
Easy sterilization
The N95 masks that health care workers wear to protect against exposure to SARS-CoV-2 and other viruses are made from polypropylene fibers that are specially designed to filter out tiny viral particles. Ideally, a health care worker would switch to a new mask each time they see a different patient, but shortages of these masks have forced doctors and nurses to wear them for longer than they are meant to be worn.
In recent months, many hospitals have begun sterilizing N95 masks with hydrogen peroxide vapor, which can be used up to 20 times on a single mask. However, this process requires specialized equipment that is not available everywhere, and even with this process, one mask can be worn for only a single day.
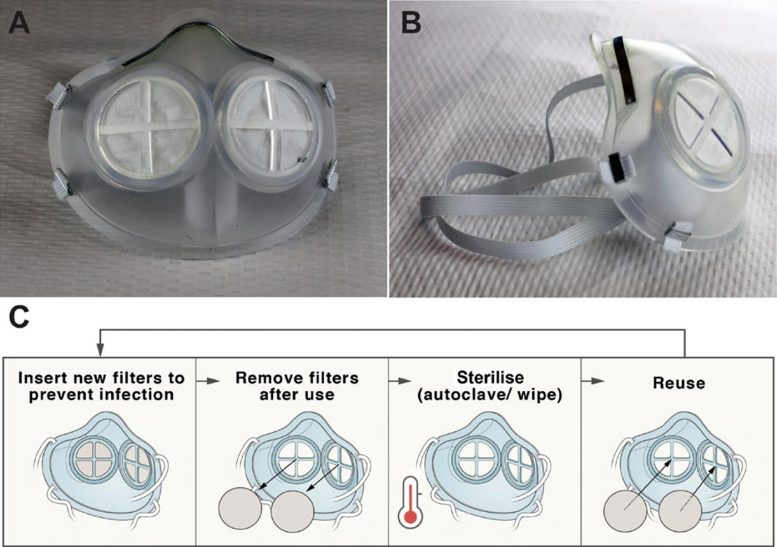
The MIT/BWH team set out to design a mask that could be safely sterilized and reused many times. They decide on silicone rubber — the material that goes into silicone baking sheets, among other products — because it is so durable. Liquid silicone rubber can be easily molded into any shape using injection molding, a highly automated process that generates products rapidly.
The masks are based on the shape of the 3M 1860 style of N95 masks, the type normally used at Brigham and Women’s Hospital. Most of the mask is made of silicone rubber, and there is also space for one or two N95 filters. Those filters are designed to be replaced after every use, while the rest of the mask can be sterilized and reused.
“With this design, the filters can be popped in and then thrown away after use, and you’re throwing away a lot less material than an N95 mask,” Wentworth says.
The researchers tested several different sterilization methods on the silicone masks, including running them through an autoclave (steam sterilizer), putting them in an oven, and soaking them in bleach and in isopropyl alcohol. They found that after sterilization, the silicone material was undamaged.
Fit test
To test the comfort and fit of the masks, the researchers recruited about 20 health care workers from the emergency department and an oncology clinic at Brigham and Women’s Hospital. They had each of the subjects perform the standard fit test that is required by the Occupational Safety and Health Administration (OSHA) for N95 masks. During this test, the subject puts the mask on and then performs a series of movements to see if the mask stays in place. A nebulized sugar solution is sprayed in the room, and if the subject can taste or smell it, it means the mask is not properly fitted.
All 20 subjects passed the fit test, and they reported that they were able to successfully insert and remove the N95 filter. When asked their preference between the new mask, a typical N95 mask, and a standard surgical mask, most either said they had no preference or preferred the new silicone mask, Byrne says. They also gave the new mask high ratings for fit and breathability.
The researchers are now working on a second version of the mask, which they hope to make more comfortable and durable. They also plan to do additional lab tests measuring the masks’ ability to filter viral particles.
As many regions of the United States have seen a surge in COVID-19 cases over the past month, hospitals in those areas face the possibility of mask shortages. There is also a need for more masks in parts of the world that don’t have the equipment needed for hydrogen peroxide sterilization.
“We know that Covid is really not going away until a vaccine is prevalent,” Byrne says. “I think there’s always going to be a need for masks, whether it be in the health care setting or in the general public.”
Reference: “Injection Molded Autoclavable, Scalable, Conformable (iMASC) system for aerosol-based protection: a prospective single-arm feasibility study” by James D Byrne, Adam J Wentworth, Peter R Chai, Hen-Wei Huang, Sahab Babaee, Canchen Li, Sarah L Becker, Caitlynn Tov, Seokkee Min and Giovanni Traverso, 7 July 2020, British Medical Journal Open.
DOI: 10.1136/bmjopen-2020-039120
The research was funded, in part, by the Prostate Cancer Foundation, the MIT Department of Mechanical Engineering, Brigham and Women’s Hospital, the National Institutes of Health, E-Ink Corporation, Gilead Sciences, Philips Biosensing, and the Hans and Mavis Lopater Psychosocial Foundation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
25 Comments
I want a mask manufacturer machine.
Let me guess, you’re a millennial
I want better comments. You know the ones that educate you and make sense? I think that before getting a mask machine one might consider learning posting on a technical website. oh yes in my pony is rainbow colored although that has nothing to do with this either dot-dot-dot
Love it!! Couldn’t of said it better myself.
Wow can i have one of that mask.
Im a frontliner here in cebu city. Mask is very essential to my life now.
Thank you!
We already have designed such masks in India, and manufacturing and delivering to Doctors and front line workers. scubemask fingurus
We already have designed such masks in India, and manufacturing and delivering to Doctors and front line workers. scubemask fingurus
I have a box of N95 masks, and it says on the back that the mask doesn’t protect the wearer from viruses. How is this mask different?
What exactly does it say? If it’s a NIOSH certified N95, then it should provide excellent protection provided it’s been properly fitted (at least do a seal check- you should instructions included with NIOSH N95, or on the NIOSH website). NIOSH isn’t part of the FDA, and the maker may not have gone through the FDA clearance which is needed to make medical claims.
If it has an exhalation valve, it might not protect others from you. Most existing elastomeric halfmask respirators (N95 to P100 levels of protection) like this one have exhalation valves, and even though they don’t have the full FDA clearance they’ve been used in hospitals for about a decade now.
D. Gonzalez, the “does not protect from viruses” is just for legal liability protection. It actually does protect from viruses, just maybe not 100%, and the company does not want to be sued in case you expect it to protect you 100% and you get sick.
Shop masks online from a huge collection at the best prices from Bewakoof Reusable Washable Pollution Mask All Weather Friendly Free Shipping COD
I would like the the website for the masks please.
If you want to test the efficacy of N95 masks with valves, try putting one on and blowing out a candle two inches in front of you. They don’t meet certain technical requirements but are orders of magnitude better at protecting others than bandanas.
I would be concerned about the fit of the circular filters in the mask and leakage between the filters and the mask. It did not look very secure in the images and the ability of the mask to work correctly depends on absolutely no leakage around the filters. If they are being replaced daily the filters must be able to be replaced without any question of leakage, poor fit, or degradation of the seal over time. I also want to point out that surgical and procedural mask material has a 98% BFE and PFE and this would also be an acceptable filter material for these silicone masks for someone that is not working directly with Covid patients but wants a decent protection from aerosol-transmitted Covid-19. That is, medical staff not working with Covid patients and the general public as well. This would be especially useful if the N95 mask material is not available in a timely manner in the quantities needed. Also because this seems to be a flat filter does this filter still have the electrostatic charge that is part of the thick N95 mask material?
It seems very true that COVID 19 is really not going away so easily as per the latest and previous statistics, and there is always going to be a need for good quality masks, whether it be in health care or in the general public. As clearly mentioned in this article the comfort and fit of the masks are very important.
We need n95 masks for everyone, not just health workers because there are so many fools out there (Republicans) making the United States the most dangerous place on Earth to be.
If I heard it once I’ve heard it a million times– wearing a mask does not protect you against the virus, it protects those around you from getting a virus. And I’m aware there a limited percentage in that protection, so don’t get crazy on me. That’s such a lazy comment because if everybody wore mask then we would all be FAR MORE protected as one could be, right? Aside from cutting off all contact with the human race. So I feel like comments like that is fuel on the fire for these people who claimed wearing a mask goes against their civil rights or constitutional rights or whatever. Listen- how about a person right to live and breathe? Do these people not have a conscience? The thought that I could be asymptomatic and a carrier is enough to keep me at home and keep to myself. The few times I had to go out, you damn straight I had a mask on the whole time. I was wearing one from the get-go, there was a couple of times my husband and I went to go grab supplies from town and we were the only people wearing a mask. It’ partly common courtesy & partly common sense. And it stills my heart that so many of my fellow fellow Americans have little of both!
Applause!
I wear a mask similar to this for woodworking..works well and comfy. However, I think eye protection needs to be part of mask. It should allow person to wear eyeglasses under it. Then it would be much safer.
I want one. How do I get it?
This thing looks like it’s terrible to wear when it’s hot.
Design consideration: Since this mask is transparent, move the filters or change the shape so people can see and read the wearer’s lips.
Great idea
Great idea for masks!
I love the idea of more efficient and reusable mask, but I’m curious if the new silicone mask are recyclable or are we just creating something else to contaminate the earth with