Researchers identify molecules that help cells to pinpoint their position, allowing for the kidneys to function properly.
For the kidneys to function flawlessly, millions of cells must be arranged precisely according to a specified blueprint. Scientists at the Max Planck Institute for Heart and Lung Research in Bad Nauheim and Philipps-Üniversität Marburg have now discovered that kidney cells send signals to neighboring cells undergoing division to organize the repairing of the kidneys after acute renal failure. The findings may help in the future to improve the regeneration of damaged organs.
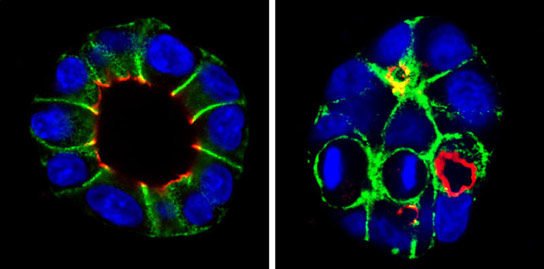
The kidneys continuously filter waste and toxic substances from the blood, which are then removed from the body via the urine. Filtration occurs in numerous microscopic tubes, known as tubules, the inner surface of which is covered by epithelial cells. Similar to boat planking, the individual epithelial cells in the tubules must be precisely arranged for the kidneys to function normally.
Intensive-care patients often suffer kidney failure, during which some of the epithelial cells die. The resulting gaps are closed by surviving epithelial cells from the surrounding area, which divide to form new cells. However, it is crucial for the new cells to be positioned at precisely the right location. If this is not the case, dividing cells could block the bore of the tubules and impair the function of the kidneys.
The spatial arrangement of the dividing epithelial cells is determined by the orientation of the spindle apparatus, a bundle of protein threads that pull the chromosomes towards the opposite poles of the cell and distributes them equally between the new cells. The Research Group led by Thomas Worzfeld at the Max Planck Institute for Heart and Lung Research in Bad Nauheim and at the Pharmacological Institute in Marburg have now investigated how the orientation of the spindle apparatus is controlled during cell division.
To this effect, the scientists from Bad Nauheim and Marburg discovered that neighboring epithelial cells communicate with each other with the help of signal molecules. In this way, they influence the orientation of the spindle apparatus and control the spatial arrangement of the new cells. “As in a GPS system, there are antennas that receive signals. This function is assumed by the plexin B2 receptor, which receives a location signal from neighboring cells,” explains Worzfeld.
The location signal itself consists of a group of signal molecules called semaphorins. Through genetic manipulation, Worzfeld’s Group switches off individual semaphorins in mice. As a result, the epithelial cells no longer arrange themselves correctly and block the tubules. “We observed the same phenomenon in mice lacking the plexin B2 receptor, meaning the antenna,” says Worzfeld.
In both cases, the damaged kidney was unable to regenerate, and it remained functionally impaired. “In this study we discovered a previously unknown function of the plexin B2 receptor and its binding partners, the semaphorins,” explains Worzfeld. It involves a fundamental mechanism by which the kidneys heal following renal failure. The scientists now want to investigate whether plexin B2 and the semaphorins also play a role in the repair of other organisms and in diseases such as cancer.
Reference: “Semaphorin-Plexin Signaling Controls Mitotic Spindle Orientation during Epithelial Morphogenesis and Repair” by Jingjing Xia, Jakub M. Swiercz, Inmaculada Bañón-Rodríguez, Ivana Matković, Giuseppina Federico, Tianliang Sun, Timo Franz, Cord H. Brakebusch, Atsushi Kumanogoh, Roland H. Friedel, Fernando Martín-Belmonte, Hermann-Josef Gröne, Stefan Offermanns and Thomas Worzfeld, 16 April 2015, Developmental Cell.
DOI: 10.1016/j.devcel.2015.02.001
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.