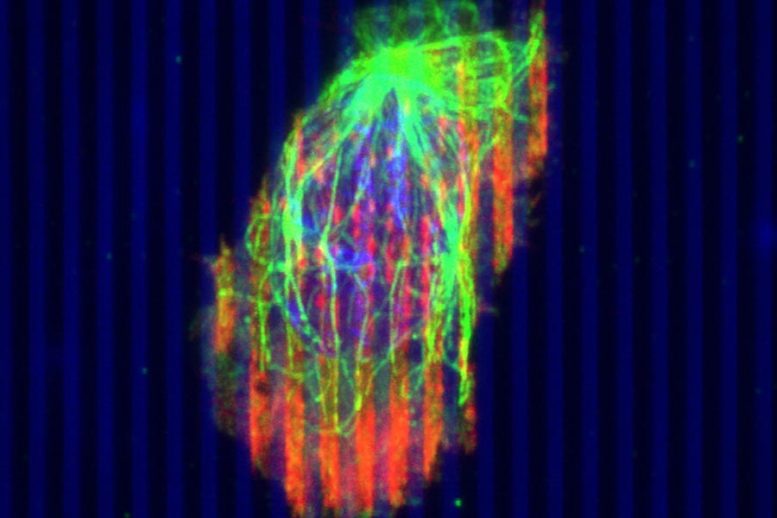
First-of-its-kind study shows how engineered immune cells move faster to attack the tumor.
A groundbreaking study led by engineering and medical researchers at the University of Minnesota Twin Cities shows how engineered immune cells used in new cancer therapies can overcome physical barriers to allow a patient’s own immune system to fight tumors. The research could improve cancer therapies in the future for millions of people worldwide.
The research is published in Nature Communications, a peer-reviewed, open access, scientific journal published by Nature Research.
Instead of using chemicals or radiation, immunotherapy is a type of cancer treatment that helps the patient’s immune system fight cancer. T cells are a type of white blood cell that are of key importance to the immune system. Cytotoxic T cells are like soldiers who search out and destroy the targeted invader cells.
While there has been success in using immunotherapy for some types of cancer in the blood or blood-producing organs, a T cell’s job is much more difficult in solid tumors.
“The tumor is sort of like an obstacle course, and the T cell has to run the gauntlet to reach the cancer cells,” said Paolo Provenzano, the senior author of the study and a biomedical engineering associate professor in the University of Minnesota College of Science and Engineering. “These T cells get into tumors, but they just can’t move around well, and they can’t go where they need to go before they run out of gas and are exhausted.”
This video shows cytotoxic T cells migrating through pancreatic tumors. University of Minnesota Twin Cities researchers are working to engineer cytotoxic T cells so they are better able to overcome the tumor’s barriers and fight cancer cells. Credit: Provenzano Group, University of Minnesota
Designing Mechanically Optimized T Cells
In this first-of-its-kind study, the researchers are working to engineer the T cells and develop engineering design criteria to mechanically optimize the cells or make them more “fit” to overcome the barriers. If these immune cells can recognize and get to the cancer cells, then they can destroy the tumor.
In a fibrous mass of a tumor, the stiffness of the tumor causes immune cells to slow down about two-fold — almost like they are running in quicksand.
“This study is our first publication where we have identified some structural and signaling elements where we can tune these T cells to make them more effective cancer fighters,” said Provenzano, a researcher in the University of Minnesota Masonic Cancer Center. “Every ‘obstacle course’ within a tumor is slightly different, but there are some similarities. After engineering these immune cells, we found that they moved through the tumor almost twice as fast no matter what obstacles were in their way.”
This high magnification video shows T cells migrating on nanopatterns engineered to mimic tumor architectures. University of Minnesota researchers are studying the mechanical properties of the cells to better understand how the immune cells and cancer cells interact. Credit: Provenzano Group, University of Minnesota
Genome Editing Boosts Immune Cell Efficiency
To engineer cytotoxic T cells, the authors used advanced gene editing technologies (also called genome editing) to change the DNA of the T cells so they are better able to overcome the tumor’s barriers. The ultimate goal is to slow down the cancer cells and speed up the engineered immune cells. The researchers are working to create cells that are good at overcoming different kinds of barriers. When these cells are mixed together, the goal is for groups of immune cells to overcome all the different types of barriers to reach the cancer cells.
Provenzano said the next steps are to continue studying the mechanical properties of the cells to better understand how the immune cells and cancer cells interact. The researchers are currently studying engineered immune cells in rodents and in the future are planning clinical trials in humans.
While initial research has been focused on pancreatic cancer, Provenzano said the techniques they are developing could be used on many types of cancers.
“Using a cell engineering approach to fight cancer is a relatively new field,” Provenzano said. “It allows for a very personalized approach with applications for a wide array of cancers. We feel we are expanding a new line of research to look at how our own bodies can fight cancer. This could have a big impact in the future.”
Reference: “Engineering T cells to enhance 3D migration through structurally and mechanically complex tumor microenvironments” by Erdem D. Tabdanov, Nelson J. Rodríguez-Merced, Alexander X. Cartagena-Rivera, Vikram V. Puram, Mackenzie K. Callaway, Ethan A. Ensminger, Emily J. Pomeroy, Kenta Yamamoto, Walker S. Lahr, Beau R. Webber, Branden S. Moriarity, Alexander S. Zhovmer and Paolo P. Provenzano, 14 May 2021, Nature Communications.
DOI: 10.1038/s41467-021-22985-5
In addition to Provenzano, the study’s authors included current and former University of Minnesota Department of Biomedical Engineering researchers Erdem D. Tabdanov (co-author), Nelson J. Rodríguez-Merced (co-author), Vikram V. Puram, Mackenzie K. Callaway, and Ethan A. Ensminger; University of Minnesota Masonic Cancer Center and Medical School Department of Pediatrics researchers Emily J. Pomeroy, Kenta Yamamoto, Walker S. Lahr, Beau R. Webber, Branden S. Moriarity; National Institute of Biomedical Imaging and Bioengineering researcher Alexander X. Cartagena-Rivera; and National Heart, Lung, and Blood Institute researcher Alexander S. Zhovmer, who is now at the Center for Biologic Evaluation and Research.
The research was funded primarily by the National Institutes of Health (NIH) and University of Minnesota Physical Sciences in Oncology Center, which receives funding from NIH’s National Cancer Institute. Additional funding was provided by the American Cancer Society and the Randy Shaver Research and Community Fund. The University of Minnesota Imaging Center provided additional staff expertise. Some of the researchers also are part of the University of Minnesota Center for Genome Engineering and the University’s Institute for Engineering in Medicine.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.