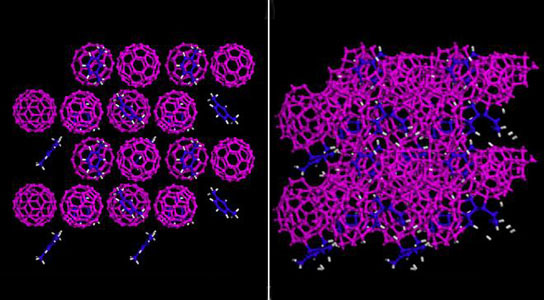
An international team of scientists created the world’s first hybrid crystalline/amorphous material that is hard enough to indent diamonds. Using a diamond anvil cell, the scientists squeezed a mixture of soccer-ball-shaped carbon-60 molecules (“buckyballs”) and a xylene solvent to pressures up to 600,000 times atmospheric pressure.
A team of scientists led by Carnegie’s Lin Wang has observed a new form of very hard carbon clusters, which are unusual in their mix of crystalline and disordered structure. The material is capable of indenting diamond. This finding has potential applications for a range of mechanical, electronic, and electrochemical uses. The work is published in the journal Science on August 17.
Carbon is the fourth-most-abundant element in the universe and takes on a wide variety of forms—the honeycomb-like graphene, the pencil “lead” graphite, diamond, cylindrically structured nanotubes, and hollow spheres called fullerenes.
Some forms of carbon are crystalline, meaning that the structure is organized in repeating atomic units. Other forms are amorphous, meaning that the structure lacks the long-range order of crystals. Hybrid products that combine both crystalline and amorphous elements had not previously been observed, although scientists believed they could be created.

Wang’s team—including Carnegie’s Wenge Yang, Zhenxian Liu, Stanislav Sinogeikin, and Yue Meng—started with a substance called carbon-60 cages, made of highly organized balls of carbon constructed of pentagon and hexagon rings bonded together to form a round, hollow shape. An organic xylene solvent was put into the spaces between the balls and formed a new structure. They then applied pressure to this combination of carbon cages and solvent, to see how it changed under different stresses.
At relatively low pressure, the carbon-60’s cage structure remained. But as the pressure increased, the cage structures started to collapse into more amorphous carbon clusters. However, the amorphous clusters still occupy their original sites, forming a lattice structure.
The team discovered that there is a narrow window of pressure, about 320,000 times the normal atmosphere, under which this new structured carbon is created and does not bounce back to the cage structure when pressure is removed. This is crucial for finding practical applications for the new material going forward.
This material was capable of indenting the diamond anvil used in creating the high-pressure conditions. This means that the material is superhard.
If the solvent used to prepare the new form of carbon is removed by heat treatment, the material loses its lattice periodicity, indicating that the solvent is crucial for maintaining the chemical transition that underlies the new structure. Because there are many similar solvents, it is theoretically possible that an array of similar, but slightly different, carbon lattices could be created using this pressure method.
“We created a new type of carbon material, one that is comparable to diamond in its inability to be compressed,” Wang said. “Once created under extreme pressures, this material can exist at normal conditions, meaning it could be used for a wide array of practical applications.”
Reference: “Long-Range Ordered Carbon Clusters: A Crystalline Material with Amorphous Building Blocks” by Lin Wang, Bingbing Liu, Hui Li, Wenge Yang, Yang Ding, Stanislav V. Sinogeikin, Yue Meng, Zhenxian Liu, Xiao Cheng Zeng and Wendy L. Mao, 17 August 2012, Science.
DOI: 10.1126/science.1220522
Wang’s other co-authors on the paper were Bingbing Liu of Jilin University, Hui Li and Xiao Cheng Zeng of the University of Nebraska, Yang Ding of the Argonne National Laboratory, and Wendy Mao of Stanford University.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.