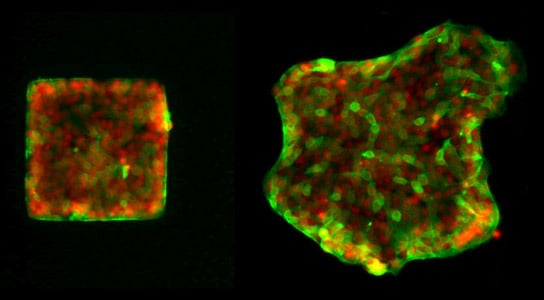
Shining trail of cell movement: Researchers at the Max Planck Institute for Intelligent Systems in Stuttgart tag the nuclei of cells (red) and the protein merlin (green) with fluorescent molecules. They then observe how merlin spreads in the cells when these migrate from a square-shaped area. As a result, they have clarified the role that merlin plays in cell movement, which is also important for wound healing. Credit: MPI for Intelligent Systems
New research from the Max Planck Institute details how the protein merlin regulates collective cell movement, promoting effective and rapid wound healing.
Cells also follow a herd instinct, and they thereby communicate in a magical collective way. This is because a protein known as merlin, named after the mythical wizard of medieval England, plays an important role in collective movement of cells in the body. This discovery was made by a team headed by scientists from the Max Planck Institute for Intelligent Systems in Stuttgart working in collaboration with University Hospital Heidelberg. In a cell, merlin registers whether and in what direction a neighboring cell is moving and induces its own cell to creep in the same direction. It is important that cells move as a cohesive group so that wounds heal quickly and effectively, for example. If this group dynamic is disrupted, however, wounds are not only closed more poorly, cancer cells can also spread more easily in the body or embryos may be stunted in their development.
The head does not always tell the body what’s going on. Skin cells, for instance, find the right way themselves. If there is a tear in the skin, the cells that are closest to the wound migrate to the gap and their neighbors simply go along. At least this is how it happens when merlin performs its role properly. If it doesn’t, the individual cells stray in a disoriented fashion through the tissue. Potentially, this could even lead to a particularly noticeable scar or the wound might not heal at all. And if the rogue cells happen to be tumor cells, it is easier for them to spread throughout the body, resulting in the formation of metastases. “Individual cells that move randomly – in other words in those cells where the tumor-inhibiting protein merlin is at least partially switched off or is not present – can enter the tissue more easily than as an orderly group of several cells,” says Joachim Spatz, Director at the Max Planck Institute for Intelligent Systems and head of the current study.
One cell can pull a series of ten other cells with it
Researchers in molecular medicine already knew that merlin inhibits the spread of tumor cells. Joachim Spatz and his team have now explained why this is so. The protein actually acts as a brake. In resting cells, merlin sits in the cortex of the cell, a protein-rich layer on the inside of the cell membrane, and more specifically on a protein complex above which neighboring cells converge. As long as merlin docks there, it also inhibits the protein Rac1, which moves a cell. If a cell moves, it pulls on the neighboring cell, or more precisely, on the protein complex to which merlin is linked. This results in a mechanical stress, which releases merlin from its parked position in the cortex. The protein then migrates into the cell plasma and no longer blocks Rac1. The cell brake is thereby released: the Rac1 protein now forms so-called lamellipodia towards the precursor cell projections. These projections pull the cell body behind them. In this way, one cell can lead and pull ten to twenty other cells that are behind it.
Researchers working with Joachim Spatz discovered that this biochemical and mechanical mechanism controls the herd instinct of cells while they were investigating the movement of skin cells. To this end, they tagged the merlin in the test cells with fluorescent proteins so that they could track its route through the respective cell.
As the researchers now have a better understanding of how cells move as a group and when they go their own way, this could also result in new approaches in medicine. The findings made by the Stuttgart-based researchers provide clues as to how impairments in wound healing could be eliminated or how the metastasis of tumors could be reduced. They also help developmental biologists in understanding how cells find their intended location in an embryo. After all, such processes are not controlled by magical powers but by the interaction of biochemistry and mechanical forces.
Reference: “A molecular mechanotransduction pathway regulates collective migration of epithelial cells” by Tamal Das, Kai Safferling, Sebastian Rausch, Niels Grabe, Heike Boehm and Joachim P. Spatz, 23 February 2015, Nature Cell Biology.
DOI: 10.1038/ncb3115





And here we have possibly got the reason why ulcers and wounds take longer to cure in warm, humid climates- merlin functions less well then?
This is very interesting stuff.