New research reveals the dual nature of supercritical fluids, transitioning from gas-like to liquid-like behavior, providing insights into fluid dynamics under extreme conditions.
A study brings remarkable insights into the enigmatic behavior of supercritical fluids, a hybrid state of matter occupying a unique space between liquids and gases, and arising in domains that go from the pharmaceutical industry to planetary science. The obtained results are at the limit of current experimental possibilities and could only be obtained in a high flux neutron source such as the Institut Laue-Langevin (ILL).
Supercritical Fluids
A liquid or gaseous substance pushed beyond its critical point (i.e., beyond the temperature and pressure at which the distinction between liquid and gas can no longer be made) is called a supercritical fluid. Still little-known and defying conventional classifications, supercritical fluids possess the ability to effuse like a gas while dissolving materials like a liquid. This duality has made them invaluable in a myriad of industrial applications, from pharmaceutical processing to decaffeinating coffee beans. On the other hand, they are crucial to understanding giant planets such as Jupiter and Neptune, where similar states of matter may reign.
Groundbreaking Research on Supercritical Fluids
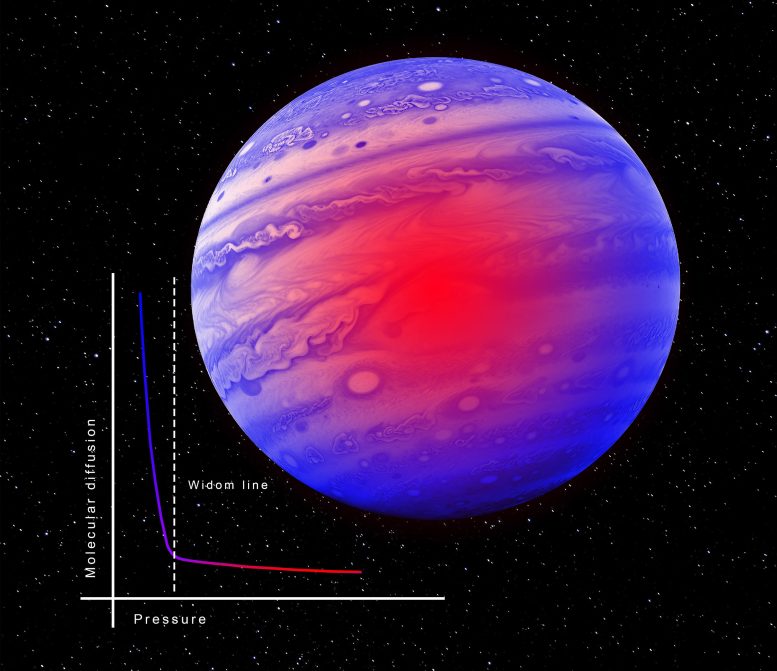
In a study published on May 16 in the journal Nature Communications, an international team of researchers from Sapienza University (Rome, Italy) Institute Laue Langevin (Grenoble, France), Ecole Polytechnique Federal (Lausanne, Switzerland), CNRS (France) and CNR (Italy) obtained experimental proof that molecular diffusion in a supercritical fluid switches from gaseous-like behavior to liquid-like behavior across the so-called Widom line (a thermodynamic line that extends the saturated vapor curve above the critical point). The transition is gradual within a narrow pressure range.
The team investigated the diffusion of molecules within a supercritical fluid – a crucial parameter reflecting the mobility of molecules within the fluid – with a fundamental question in mind: can we pinpoint a region of pressure-temperature where the behavior of a supercritical fluid goes from gas-like to liquid-like? While theoretical models have proposed various such transition boundaries (among them the Widom line) experimental validation had remained elusive, until now.
Experimental Insights Into Molecular Diffusion
This result was obtained through challenging, high-pressure, quasi-elastic neutron scattering (QENS) experiments on supercritical methane conducted at the Institut Laue Langevin, in Grenoble. At the ILL, neutrons are used to explore materials and processes in all possible ways in a very wide range of domains. In this study, a neutron beam was sent onto a cell containing methane in supercritical conditions.
The intensity of the neutron beam scattered by the sample was measured as a function of the energy exchanged in the range of interest (i.e., in the energy range where molecular diffusion phenomena within matter occur, the so-called quasi-elastic regime). The measurements took place at constant temperature T=200 K (above the critical T=190 K) varying the pressure of the methane from a few bars up to very high pressures (reaching nearly 3 Kbar; the critical pressure is P=45 bar). The experiments were conducted on ILL instrument IN6-SHARP.
The authors underline the strikingly clear experimental evidence: “While at pressures lower than approximately 50 bar the signal of the diffusion dynamics typical of gaseous systems is observed, we have been able to observe that as the pressure increases above that, the signal evolves progressively until it takes on the typical shape of liquids” explains author Alessio De Francesco (researcher at CNR and ILL).
The result was made possible thanks to the high flux neutron source and the unique experimental support facilities available at the ILL: “These measurements are at the limits of current experimental possibilities, and were unthinkable until a few years ago” adds Ferdinando Formisano (researcher at CNR and ILL), concluding: “As often happens in research, having opened a door means seeing new avenues to explore, and this objective can only be pursued thanks to access to large research facilities.”
Reference: “Crossover from gas-like to liquid-like molecular diffusion in a simple supercritical fluid” by Umbertoluca Ranieri, Ferdinando Formisano, Federico A. Gorelli, Mario Santoro, Michael Marek Koza, Alessio De Francesco and Livia E. Bove, 16 May 2024, Nature Communications.
DOI: 10.1038/s41467-024-47961-7
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.