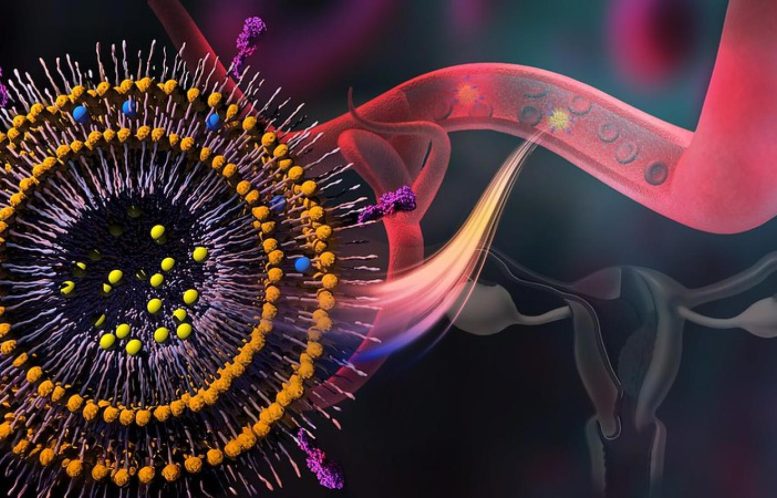
A new nanomedicine drug delivery system improves chemotherapy targeting for choriocarcinoma, shrinking tumors by 95% in mouse models. Researchers engineered polymersomes to enhance methotrexate (MTX) delivery, making treatment more effective with fewer side effects. This innovation could benefit other cancers as well.
A new drug delivery system shows promise for treating choriocarcinoma, a rare and aggressive cancer that affects pregnant women and new mothers. Researchers believe this approach could also be effective against other types of cancer.
A team led by Olena Taratula, a nanomedicine researcher at Oregon State University, has developed a method to improve drug targeting, ensuring the treatment reaches tumor cells while minimizing damage to healthy tissue.
The study, published in Small Science, focuses on choriocarcinoma, which occurs in approximately four out of every 100,000 pregnancies in the United States. This cancer typically originates in the uterus from placental cells and can develop following a miscarriage, abortion, or ectopic pregnancy, where a fertilized egg implants outside the uterine lining.
It can also happen following a molar pregnancy (no embryo forms, and the placental tissue grows abnormally) and even after a full-term pregnancy.
Targeting Tumor Cells with Nanomedicine
Taratula and collaborators including OSU postdoctoral researcher Babak Mamnoon and Maureen Baldwin, a physician at Oregon Health & Science University, designed a type of drug nanocarrier known as a polymersome to specifically target a protein in choriocarcinoma cells.
Polymersomes are hollow spheres that are synthetic versions of liposomes, lipid-based sacs found in all living cells. The protein the researchers aimed for is equilibrative nucleoside transporter 1, usually abbreviated as ENT-1, which is important for a range of cellular processes, among them DNA and RNA synthesis.
In addition to being abundant in choriocarcinoma cells, ENT-1 is found in the brain, heart, liver, and other tissues in the body.
Mamnoon led the research team in mouse model testing that confirmed attaching guanosine, a building block of RNA, to the polymersome allowed it to deliver more of the chemotherapy drug methotrexate directly to tumor cells.
“Given MTX’s role as the mainstay treatment for choriocarcinoma, the critical goal now is to enhance its effectiveness, including faster response times, while simultaneously minimizing side effects,” said Taratula, an associate professor in the OSU College of Pharmacy.
Enhancing Methotrexate Effectiveness
Methotrexate or MTX, a common cancer drug, works by interfering with cells’ ability to use folic acid, essential for making DNA and RNA. By blocking a certain enzyme, MTX thwarts replication of the otherwise rapidly dividing cancer cells.
Common symptoms of choriocarcinoma are pelvic pain and irregular vaginal bleeding. The cancer can quickly spread via the bloodstream to other parts of the body including bones, gastrointestinal tract, breasts, kidneys, liver, lungs, lymph nodes, and brain.
“Because choriocarcinoma occurs in people who were recently pregnant, they often have young families, so we need to make getting diagnosis and treatment easier and faster for them,” said Baldwin, an obstetrician/gynecologist at OHSU.
Most cases, especially if caught early, are curable, with a five-year survival rate of about 87%.
“But MTX has poor tumor specificity in standard applications and can cause side effects such as liver and kidney toxicity,” Taratula said. “That’s why we need a specially designed nanoplatform that ensures precise drug delivery and release directly into tumors.”
In the mouse model, the approach reduced tumor size by 95%, working roughly six times better than non-targeted drug carriers. The upshot is more effective treatment coupled with fewer or less severe side effects, and with further research, the same approach could be applied to treating other cancers, Taratula said.
Reference: “ENT-1-Targeted Polymersomes to Enhance the Efficacy of Methotrexate in Choriocarcinoma Treatment” by Babak Mamnoon, Ana Paula Mesquita Souza, Tetiana Korzun, Maureen K. Baldwin, K. Shitaljit Sharma, Oleh Taratula, Yoon Tae Goo, Prem Singh, Vladislav Grigoriev, Aryan Lakhanpal and Olena R. Taratula, 28 January 2025, Small Science.
DOI: 10.1002/smsc.202400361
Financial support for the study came from the OSU College of Pharmacy, the OHSU School of Medicine, the National Institutes of Health, and the Eunice Kennedy Shriver National Institute of Child Health and Human Development.
Collaborators included Oregon State’s Ana Paula Mesquita Souza, Tetiana Korzun, K. Shitaljit Sharma, Oleh Taratula, Yoon Tae Goo, Prem Singh, Vladislav Grigoriev and Aryan Lakhanpal.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.