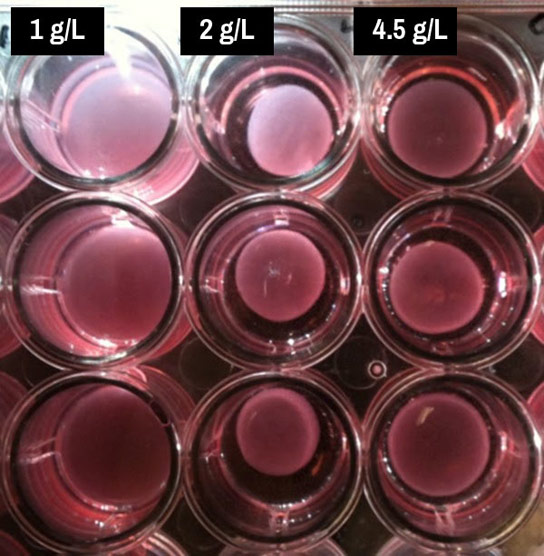
Assays created in a Rice University lab contain interstitial heart-valve cells, collagen, and nutrients. These contain varying levels of glucose metabolized by the cells as they create the extracellular matrix that forms valves. The lab found that 2 grams per liter of glucose in the solution (center vertical row) worked best, but either more or less slowed the process. Credit: Grande-Allen Lab/Rice University
By directly viewing the effect of high blood-sugar levels on heart-valve cell metabolism for the first time, bioengineers have found new evidence of a possible link between diabetes and the hardening of heart valves.
Rice University bioengineers have found new evidence of a possible link between diabetes and the hardening of heart valves.
A Rice lab, in collaboration with the University of Texas Health Science Center at Houston (UTHealth) Medical School, discovered that the interstitial cells that turn raw materials into heart valves need just the right amount of nutrients for proper metabolic function.
The surprise was that feeding them too much glucose, a sugar, slowed the cells down.
Diabetes is a metabolic disease characterized by high blood-sugar levels over a long period; a 2006 study of atherosclerosis by University of Washington researchers found a correlation between diabetes and aortic-valve calcification.
In the new work by the lab of Jane Grande-Allen of Rice’s bioengineering department, recently ranked No. 5 in the nation by U.S. News & World Report, researchers have directly seen the effect of high blood-sugar levels on heart-valve cell metabolism for the first time. The study appears this month in the Journal of the Royal Society Interface.
“The most significant result of the study is that high glucose concentration can actually be detrimental to the aortic-valve cells and their behavior in interacting with the extracellular matrix,” said lead author Peter Kamel, who carried out the experiments as a Rice undergraduate. He is in his third year at Baylor College of Medicine, where he is completing a dual-degree program offered by the neighboring Houston institutions.
“We’ve seen in a variety of other cell types, like cells in the kidney, the retina, and nerves, that high glucose concentrations can directly damage those cells and their activities,” he said. “That results in patients with diabetes having problems with vision and with their nerves and kidneys as well.
“The results that high glucose concentration can also cause pathologic remodeling by the aortic-valve cells could suggest that diabetes is also directly a cause of aortic-valve disease,” he said.
Grande-Allen’s lab studies the biomechanics of heart valves, particularly their calcification, or hardening, a condition that lessens blood flow to the heart. In the new study the researchers took on the little-understood process of interstitial valve-cell metabolism.
“Hardly anything was known about the metabolism of heart valves, but metabolism underlies everything,” Grande-Allen said. “It’s one of the cell’s main orders of business.”
The idea for the study came about when she met with Heinrich Taegtmeyer, a cardiologist and professor at UTHealth. Kamel’s experiments combined Taegtmeyer’s ideas about cell metabolism and the Grande-Allen lab’s capabilities to explore ways to manipulate interstitial cells. Rice chemical engineer Deepak Nagrath also contributed key insights about how to examine the metabolism of these cells.
Kamel started with solutions of collagen, the most abundant protein in the body, and cells drawn from the aortic heart valves of pigs. Most solutions also included a nutrient — glucose, pyruvate, glutamine, or a supplement mixture called F-12 — to stimulate the cells’ metabolism.
When the cells worked at peak, Grande-Allen said, they would contract the liquid into a gel as they absorbed and metabolized nutrients, and then turned raw collagen into connective tissue. “The cells pulled the collagen together and tightened it into packed structures,” she said.
“This test is extremely sensitive to how much glucose you give the cells,” she said. “When we didn’t give them any glucose, nothing happened. If we gave them just a little, we would wait and wait. Sometimes, after about two weeks, the cells would start contracting.
“Glucose helps cells do many things. It provides essential fuel for their function, and it helps them make carbohydrates that are part of glycoproteins and proteoglycans and glycolipids, but the cells have to work really hard to process all that,” Grande-Allen said. “Maybe that’s what’s happening to the collagen gel contraction when we give them more glucose.”
The researchers found solutions with 2 grams of glucose per liter (0.27 ounces per gallon)were optimal for contraction. Because off-the-shelf solutions come in standard concentrations of 1 and 4.5 grams per liter (0.13 and 0.6 ounces per gallon), researchers who don’t mix their own solutions could easily miss the connection between glucose level and metabolism, Kamel said.
“It’s interesting that the standard concentrations weren’t the best,” Grande-Allen said.
“I also want to follow up on the effects of higher glucose concentrations,” she said. “We know that calcific aortic-valve disease is heavily associated with metabolic syndrome, which can lead to Type 2 diabetes. It’s not as common as the link between atherosclerosis and diabetes, but it’s definitely an appreciable, strong subset.”
To understand any heart valve-related disease, “we need to understand the mechanism of how the cells interact with excess lipids and sugars, and we’re just starting to scratch the surface,” she said. “So work like this is pretty fundamental.”
Co-authors are former Rice postdoctoral researcher Xin Qu, now a postdoctoral fellow at the University of California, San Diego; Rice graduate student Andrew Geiszler; Nagrath, an assistant professor of chemical and biomolecular engineering at Rice; and Romain Harmancey, a postdoctoral fellow, and Taegtmeyer, a professor of medicine at The University of Texas Medical School at Houston. Grande-Allen is a professor of bioengineering at Rice.
The National Institutes of Health, the Rice Century Scholars Program, and a Hamill Innovation Award by the Rice University Institute of Biosciences and Bioengineering supported the research.
Reference: “Metabolic regulation of collagen gel contraction by porcine aortic valvular interstitial cells” by Peter I. Kamel, Xin Qu, Andrew M. Geiszler, Deepak Nagrath, Romain Harmancey, Heinrich Taegtmeyer and K. Jane Grande-Allen, 6 December 2014, Journal of the Royal Society Interface.
DOI: 10.1098/rsif.2014.0852








HbA1c test is already there to calculate the glucose concentration average for the past 3 months,by deciding on the amount of glucose sticking in the blood red corpuscles when they are in circulation and particularly when they are hyperglycemic. So, it is natural that residual glucose should be sticking in the other organs like kidney, eye retina, heart valve etc., as long as the sugar is not under control. Just like the fibrinogen which are broken platelets weaving the hardening of injured tissues, so much so this process of excess sugar sticking constantly to the heart valves if the sugar is not under control can contribute for the hardening of the arteries. if the sugar is brought under control continuously then the sticky proteins can be flushed out slowly. thank You.