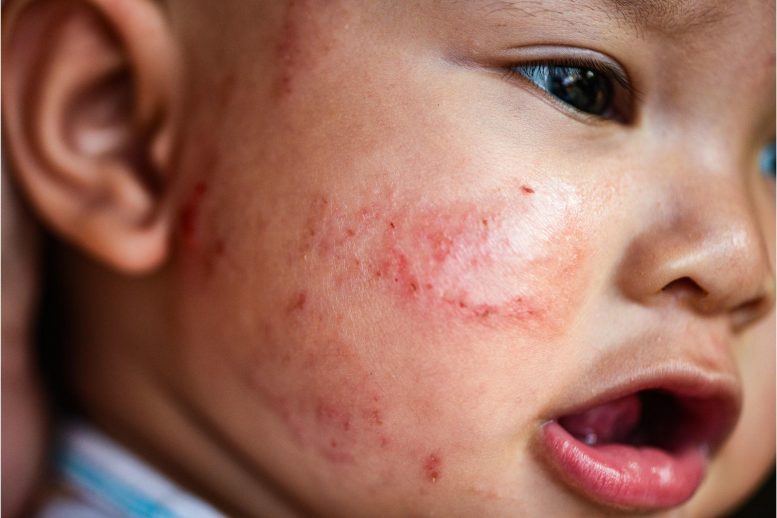
Dupilumab, a biologic drug, reduces severe eczema symptoms safely in young children and may prevent associated allergies.
Researchers involved in a new multi-site international phase III study led by Northwestern Medicine found that dupilumab was highly effective in reducing the signs and symptoms of moderate-to-severe eczema. This is the first study to treat moderate-to-severe eczema in infants and children 6 months to 5 years of age with a biologic drug (monoclonal antibody) instead of immune-suppressing medications.
More than half the children saw at least a 75% decrease in signs of eczema, highly significant reductions in itching, and better sleep after a 16-week course of dupilumab, a medication that targets a critical immune pathway in allergies.
This is the first large, random, placebo-controlled study of a monoclonal antibody for any skin condition, including eczema, in children as young as six months. The research, which included 31 sites throughout Europe and North America, was recently published in the journal The Lancet.
“Preschoolers who are constantly scratching, awake multiple times a night with their parents, irritable and markedly curtailed in their ability to do what other children their ages can do improved to the extent that they sleep through the night, change their personalities and have a normal life — as babies and children should,” said lead study author Dr. Amy Paller, chair of dermatology at Northwestern University Feinberg School of Medicine and an attending physician at Ann & Robert H. Lurie Children’s Hospital of Chicago.
Impact of Eczema on Childhood Development and Sleep
Eczema, also known as atopic dermatitis, is a chronic inflammatory skin condition that causes itching and red, dry, often oozing, skin. It can have a significant impact on the life of both the patient and their family.
About one-fifth of all children under the age of six are thought to have eczema and 85 to 90% of those who have the condition as a whole experience its onset in the first five years of life.
The children’s debilitating itch causes sleep problems, poor neurocognitive development, and, on average, a full night of missed sleep per week.
“The ability to take this drug will significantly improve the quality of life for infants and young children who suffer tremendously with this disease,” Paller said. “Atopic dermatitis or eczema is so much more than just itchy skin. It is a devastating disease. The quality of life of severe eczema — not only for the child but also parents — is equivalent to many life-threatening diseases.”
As a result of this study, this medication is now available to infants and preschoolers as young as 6 months of age. It has “an outstanding safety profile” and does not even require any laboratory tests before starting the medication, Paller said.
Although one-half to two-thirds of young children with eczema have mild symptoms, which can be handled with steroid ointment and moisturizers, the other one-third or more have moderate-to-severe disease and require more aggressive management.
Dupilumab: A Safe Alternative to Immunosuppressive Drugs
“Up to now, all we have had to treat more severe eczema is immune-suppressing medications, such as oral steroids, which we try to avoid in children because they are associated with so many side effects and thus are not a preferred treatment for a chronic skin disease,” Paller said. “The potential long-term impact on the development of the immune system in young children is also of concern with these immunosuppressants.”
During the past few years, a new medication has become available called dupilumab, which is the first “biologic” drug to treat eczema in a targeted manner, meaning a narrow attack on just what scientists have found is causing the manifestations of the disease in the skin. This medication was found to be effective and safe in studies with adults, then adolescents, then other school-aged children.
“But the group in whom we worry the most about safety — those under 5 — had not been tested and were unable to get this medication,” Paller said.
The parent or a health care provider gives the child a monthly shot to administer the medication.
“The effect for most of these younger children is dramatic and at least as good as we’ve seen with the risky immunosuppressant medications,” Paller said.
Potential Added Benefit by Treating Associated Allergies
This medication has also been shown to be effective for treating asthma, gastrointestinal manifestations of allergy, and other allergy-mediated problems but is not yet approved for these indications in infants and young children.
In fact, 66% of children in this trial had developed their eczema during the first six months of life and, by the time of initiating the dupilumab, more than 80% had already developed at least one allergic disorder, such as asthma or food allergy.
“By treating more aggressively to calm the immune system activation in these young children with early, severe eczema, we may also reduce the risk of their developing a range of allergic problems, changing their life beyond improving eczema,” Paller said. “These associated allergic issues most often begin after eczema starts.”
Children were randomized to receive either a placebo injection or the dupilumab (weight-based dosing) every four weeks for 16 weeks. Only children who were not responding adequately to topical medications were allowed to enroll, and they had to be of high severity, even with the topical medications.
As a result of the study, Paller said, scientists and physicians can start to better understand the relationships between eczema and a variety of allergic disorders and can consider the possibility of using this medication for other disorders that affect these very young children.
Reference: “Dupilumab in children aged 6 months to younger than 6 years with uncontrolled atopic dermatitis: a randomised, double-blind, placebo-controlled, phase 3 trial” by Amy S Paller, MD, Eric L Simpson, MD, Elaine C Siegfried, MD, Michael J Cork, MD, Andreas Wollenberg, MD, Peter D Arkwright, MD, Weily Soong, MD, Mercedes E Gonzalez, MD, Lynda C Schneider, MD, Robert Sidbury, MD, Benjamin Lockshin, MD, Steven Meltzer, MD, Zhixiao Wang, Ph.D., Leda P Mannent, MD, Nikhil Amin, MD, Yiping Sun, Ph.D., Elizabeth Laws, Ph.D., Bolanle Akinlade, MD, Myles Dillon, Ph.D., Matthew P Kosloski, Ph.D., Mohamed A Kamal, PharmD, Ariane Dubost-Brama, MD, Naimish Patel, MD, David M Weinreich, MD, George D Yancopoulos, MD, John T O’Malley, MD and Ashish Bansal, MD, on behalf of the participating investigators, 17 September 2022, The Lancet.
DOI: 10.1016/S0140-6736(22)01539-2
The clinical trial was funded by Regeneron Pharmaceuticals, Inc. and Sanofi, who jointly developed dupilumab.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
14 Comments
I had severe eczema when I was little and it got so bad I would use a wire hairbrush to scratch my flesh. Sometimes it tore my skin off. Then one of my mom’s hippy friends told her to mix safflower oil in a big glass of orange juice. After drinking this concoction (tasted horrible but so worth it) I never dealt with eczema again. Best of luck to everyone who suffers from this nightmare. Always try natural remedies prior to a pharmaceutical prescription. This is certain.
My eczema returned when i turned 70 leaving pigmentation loss with white spots. If sonething biologic can work on kids i would try it also.
I want to know medicines name.i suffering eczema.
I wish we could see studies being done at Northwestern on red skin syndrome/TSW topical steroid withdrawal. These patients are suffering terrible iatrogenic disease from prescribed steroids for eczema.
My son 7 years old it started excema on his lower area of his lips. The Dr. recommended hydrocortisone cream. I applied everytime when the weather changes. Special in winter he gets rush and very bad red spot. He is leaking his lips like a puppy. So sad, to see your kid doing that. Seems the hydrocortisone is really helping for long. What is the name of this medication. It will lovely is one of this reporter telling the information about it.
My son is 13 now and he still has it very bad he has also developed food allergies and other allergies as well non of the medication steroids has worked for him how can I get some of this medication
My daughter was diagnosed at the age of 2 but symptoms appear before diagnosis she is 11 now and 2022 had been our worst year normally by spring eczema would clear but from May-November it’s been so servere never seen it this bad she experienced flaking,bleeding ,pain swelling skin pulling according to her and wakes about 3 times a night it’s been exhausting.Please if this medication can help it would be a God sent.
My son is 9yrs he had it since birth. He can’t sleep at all. He scratches his body all day long. Pls help with the name of the medicine.
Very possible causes start with mothers diet while pregnant. Sensitivity is started then with addition of soy based formula the cascade of reactions start. Soy in the US is contaminated with pesticides. These accumulate and start an overactive immune response.
Dupilumab is the generic name of the medication in this article. Dupixent is the brand name of the medication. Since it is a biologic drug it is super expensive. Either you have good drug coverage or you are a rockstar and are rich enough to afford it. Otherwise you will not be able to buy the medication so it may as well not exist thanks to corporate greed.
It’s amazing how we always try to work on the symptoms, usually with some drug 85% of our problems are diet related. Eczema is mostly due to dairy products.
I’ve been on dupixent for about 5 maybe 6 months now, I’ve suffered for about 15 years (I’m now 21) with my eczema…. Taking everything you can think of from creams to oral meds, and I’ve never had better results than what dupixent has given me…. I would 100% recommend getting dupixent and I would also recommend taking it in the stomach cause I found doing the shot in your thigh hurts where in the stomach doesn’t! 🙂
My son has been on this medication for 16 months and it’s completely changed his and our lives. He was diagnosed with behaviour issues, ADHD and was struggling in school. About 6 months after he started Dupixent, he was happy at school, able to participate and his behaviour issues disappeared. We took him off his ADHD meds and things only continued to improve from there. He’s catching up academically at a pretty impressive rate. People don’t truly understand how much eczema impacts everyday life. This medication is amazing and I will forever be singing it’s praises!
If you can get valisone, it works. I got it in Des Moines, Iowa, but moved to Fl. Unable to get it here, probably because it works.