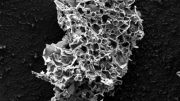
A synthesis procedure developed by NITech scientists can convert fish scales obtained from fish waste into a useful carbon-based nanomaterial. Their approach uses microwaves to break the scales down thermally via pyrolysis in less than 10 seconds, yielding carbon nano-onions with unprecedented quality compared with those obtained from conventional methods. Credit: Takashi Shirai from NITech, Japan
Researchers create a simple, quick, and energy-efficient approach for synthesizing quality carbon nano-onions from fish scales.
Carbon-based nanomaterials are increasingly being used in electronics, energy conversion and storage, catalysis, and biomedicine due to their low toxicity, chemical stability, and extraordinary electrical and optical properties. CNOs, or carbon nano-onions, are by no means an exception. CNOs, which were first described in 1980, are nanostructures made up of concentric shells of fullerenes that resemble cages inside cages. They have several desired qualities, including a large surface area and high electrical and thermal conductivities.
Unfortunately, there are also significant disadvantages to using conventional methods to produce CNOs. Some call for harsh synthesis conditions, including high temperatures or vacuum, while others demand a great deal of time and energy. While certain methods may get beyond these limitations, they still need complicated catalysts, expensive carbon sources, or potentially hazardous acidic or basic conditions. This severely restricts CNOs’ potential.
Scientists have developed a simple, fast, and energy-efficient synthesis method for producing exceptional carbon nano-onions from fish scales. Credit: Takashi Shirai from NITech, Japan
Fortunately, there is still hope. A group of researchers from Nagoya Institute of Technology in Japan recently discovered a simple and easy method to convert fish waste into extremely high-quality CNOs. Their findings were recently published in the journal Green Chemistry. The group, which included Associate Professor Takashi Shirai, Master’s student Kai Odachi, and Assistant Professor Yunzi Xin, created a method of synthesis in which fish scales, which are extracted from fish waste after cleaning, are quickly converted into CNOs by microwave pyrolysis.
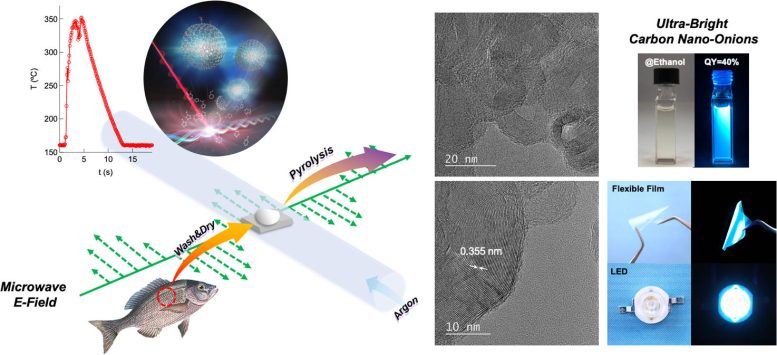
(Left) Scheme depicting the synthesis of carbon nano-onions via the microwave pyrolysis of fish scales. The top inset shows the rise in temperature of the fish scales due to microwave absorption over a period of 10 seconds, as well as a proposed formation mechanism for the carbon nano-onions. (Right) transmission electron microscopy images showing the morphology of the synthesized carbon nano-onions and photographs of CNO dispersion in ethanol, an emissive flexible film, and an LED containing CNO. Credit: Takashi Shirai from NITech, Japan
But how can fish scales be converted into CNOs so easily? While the exact reason is not altogether clear, the team believes that it has to do with the collagen contained in fish scales, which can absorb enough microwave radiation to produce a fast rise in temperature. This leads to thermal decomposition or “pyrolysis,” which produces certain gases that support the assembly of CNOs. What is remarkable about this approach is that it needs no complex catalysts, harsh conditions, or prolonged wait times; the fish scales can be converted into CNOs in less than 10 seconds!
Moreover, this synthesis process yields CNOs with very high crystallinity. This is remarkably difficult to achieve in processes that use biomass waste as a starting material. Additionally, during synthesis, the surface of the CNOs is selectively and thoroughly functionalized with (−COOH) and (−OH) groups. This is in stark contrast to the surface of CNOs prepared with conventional methods, which is typically bare and has to be functionalized through additional steps.
This “automatic” functionalization has important implications for the applications of CNOs. When the CNO surface is not functionalized, the nanostructures tend to stick together owing to an attractive interaction known as pi−pi stacking. This makes it difficult to disperse them in solvents, which is necessary for any application requiring solution-based processes. However, since the proposed synthesis process produces functionalized CNOs, it allows for excellent dispersibility in various solvents.
Yet another advantage associated with functionalization and high crystallinity is that of exceptional optical properties. Dr. Shirai explains: “The CNOs exhibit ultra-bright visible-light emission with efficiency (or quantum yield) of 40%. This value, which has never been achieved before, is about 10 times higher than that of previously reported CNOs synthesized via conventional methods.”
To showcase some of the many practical applications of their CNOs, the team demonstrated their use in LEDs and blue-light-emitting thin films. The CNOs produced a highly stable emission, both inside solid devices and when dispersed in various solvents, including water, ethanol, and isopropanol.
“The stable optical properties could enable us to fabricate large-area emissive flexible films and LED devices,” speculates Dr. Shirai. “These findings will open up new avenues for the development of next-generation displays and solid-state lighting.”
Furthermore, the proposed synthesis technique is environmentally friendly and provides a straightforward way to convert fish waste into infinitely more useful materials. The team believes their work would contribute to the fulfillment of several of the UN’s Sustainable Development Goals. Additionally, if CNOs make their way into next-generation LED lighting and QLED displays, they could greatly help reduce their manufacturing costs.
Let us hope the efforts of these scientists tip the scales in favor of CNOs for more practical applications!
Reference: “Fabrication of ultra-bright carbon nano-onions via a one-step microwave pyrolysis of fish scale waste in seconds” by Yunzi Xin, Kai Odachi and Takashi Shirai, 25 April 2022, Green Chemistry.
DOI: 10.1039/D1GC04785J






Be the first to comment on "New Method Converts Fish Waste Into Valuable Nanomaterial in Seconds"