
Researchers have uncovered the molecular structure of the protein UCP1, which allows brown fat tissue to burn calories as heat. This breakthrough could pave the way for treatments that artificially activate UCP1, thereby combating obesity and diabetes by burning off excess calories.
A St Catharine’s PhD student and one of the College’s alumni are part of a team of researchers at the University of Cambridge and the University of East Anglia who made an important discovery in the race to find treatments for obesity and related diseases, such as diabetes.
Scott Jones is completing his PhD at the University of Cambridge’s MRC Mitochondrial Biology Unit and is the first author of the new study, which revealed for the first time the molecular structure of a protein called ‘uncoupling protein 1’ (UCP1). This protein allows brown fat tissue, or ‘good fat’, to burn off calories as heat – in contrast to conventional white fat which stores calories.
Scott said, “Solving the structure of human uncoupling protein has been the focus of my PhD studies so I’m delighted the structure is now published allowing us to further understand how the protein works and is regulated.”
Alumnus Dr. Martin King (2002, Natural Sciences) is also based at the University of Cambridge’s MRC Mitochondrial Biology Unit and was a co-author of the paper published recently in the journal Science Advances. The breakthrough was made thanks to an international collaboration between the UK team and colleagues at the University of Pennsylvania and the Free University of Brussels.
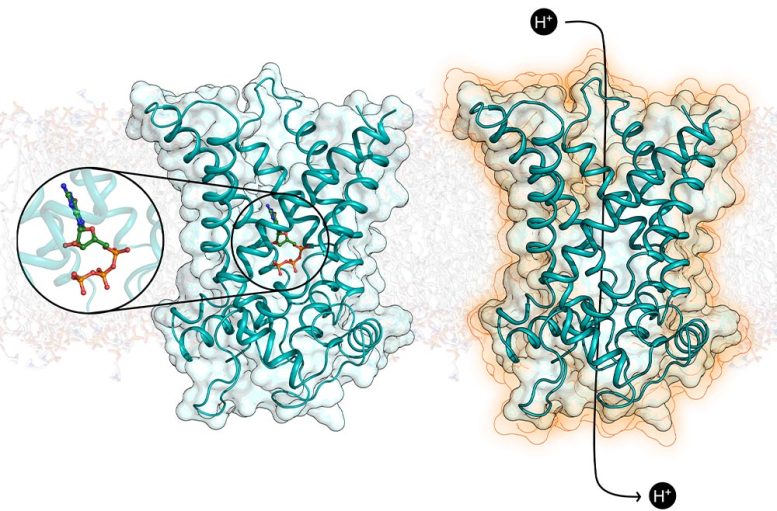
Dr. King explained, “This has been a long-term project (spanning more than 10 years) and is a great example of how fruitful collaborations in science can be. Each lab has its own expertise that has been essential in solving this problem.”
These findings provide crucial molecular details that will help develop therapeutics that activate UCP1 artificially to burn off excess calories from fat and sugar. And that this could one day combat obesity and related diseases, such as diabetes.
How UCP1 Regulates Fat Burning
Dr. Paul Crichton from the University of East Anglia said:
“As well as the conventional white fat that we are all familiar with, we can also develop brown fat. Brown fat is the good fat – it breaks down blood sugar and fat molecules to create heat and help maintain body temperature. Most of our fat, however, is white fat, which stores energy – and too much white fat leads to obesity. UCP1 is the key protein that allows the specialized brown fat to burn off calories as heat.
“We know that mammals switch on UCP1 activity in brown fat tissue to protect against the cold and to maintain body temperature – especially in new-borns, that cannot yet shiver to keep warm. Brown fat varies in humans, where it correlates with leanness in the population – and there has been a lot of interest in how to increase brown fat and activate UCP1 therapeutically, as a potential way to treat obesity.
“A lot of research has been focusing on finding ways to encourage brown fat and how to turn white fat into brown fat – in order to burn more calories and fight metabolic disease. But even with more brown fat – UCP1 must still be ‘switched on’ to gain full benefit. And research has been hampered by a lack of details on the molecular makeup of UCP1. Despite more than 40 years of research, we did not know what UCP1 looks like to understand how it works – until now.”
Using the Krios G3i, a cryogenic electron microscope at the Penn Singh Center for Nanotechnology, the team was able to view UCP1 in atomic detail.
Lead researcher from the University of Cambridge, Professor Edmund Kunji, said:
“Our paper reveals, for the first time, the structure of UCP1 in atomic detail, and how its activity in brown fat cells is inhibited by a key regulatory molecule. Our work shows how a regulator binds to prevent UCP1 activity, but more importantly, the structure will allow scientists to rationalize how activating molecules bind to switch the protein on, leading to the burning of fat. The activated tissue can also remove glucose from the blood, which can help control diabetes. This is a significant breakthrough in this field.”
For more on this research, see The Breakthrough That Could Lead to New Obesity Treatments.
Reference: “Structural basis of purine nucleotide inhibition of human uncoupling protein 1” by Scott A. Jones, Prerana Gogoi, Jonathan J. Ruprecht, Martin S. King, Yang Lee, Thomas Zögg, Els Pardon, Deepak Chand, Stefan Steimle, Danielle M. Copeman, Camila A. Cotrim, Jan Steyaert, Paul G. Crichton, Vera Moiseenkova-Bell, Edmund R. S. Kunji, 31 May 2023, Science Advances.
DOI: 10.1126/sciadv.adh4251
This research was supported by the Medical Research Council, the Biological and Biotechnological Sciences Research Council, and by the National Institutes of Health/National Institute of General Medical Sciences. Nanobody discovery was funded by the Instruct-ERIC part of the European Strategy Forum on Research infrastructures, and the Research Foundation – Flanders, and the Strategic Research Program of the Vrije Universiteit Brussel.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
Before everyone asks “How do I buy this UCP1? Put me in the study” again, they’ve learned more about the UCP1 in your body, which helps the body tell fat to make you warmer, burning more fat. They don’t know how to make that happen yet.
The best way to “fat burn” is stop eating like a pig…..there is NO excuse for being fat other than being unfortunate to be a bed bound invalid. Try a no alcohol green salad diet, no potatoes, no chips, no mcfatburgers, and even a little exercise….works wonders.