
According to research conducted by the University of Pennsylvania, the enzyme DOT1L, a stem cell self-renewal factor, is necessary for mice to continue producing sperm throughout adulthood.
Men may continue to generate sperm throughout their adult life, in contrast to women who are born with all the eggs they will ever have. To do so, they must constantly renew the spermatogonial stem cells that give birth to sperm.
According to research by Jeremy Wang of the University of Pennsylvania School of Veterinary Medicine and colleagues, this stem cell renewal is dependent on a recently identified stem cell self-renewal factor known as DOT1L. The scientists demonstrated that animals lacking DOT1L are unable to retain spermatogonial stem cells, which affects their ability to constantly make sperm.
The finding, which was reported in the journal Genes and Development, adds another entity to the handful of stem cell renewal factors that have already been identified by scientists.
“This novel factor was only able to be identified by finding this unusual phenotype: the fact that mice lacking DOT1L were not able to continue to produce sperm,” says Wang, the Ralph L. Brinster President’s Distinguished Professor at Penn Vet and a corresponding author on the paper.
“Identifying this essential factor not only helps us understand the biology of adult germline stem cells but could also allow us to one day reprogram somatic cells, like a type of skin cell called fibroblasts, to become germline stem cells, essentially creating a gamete in a petri dish. That is the next frontier for fertility treatment.”
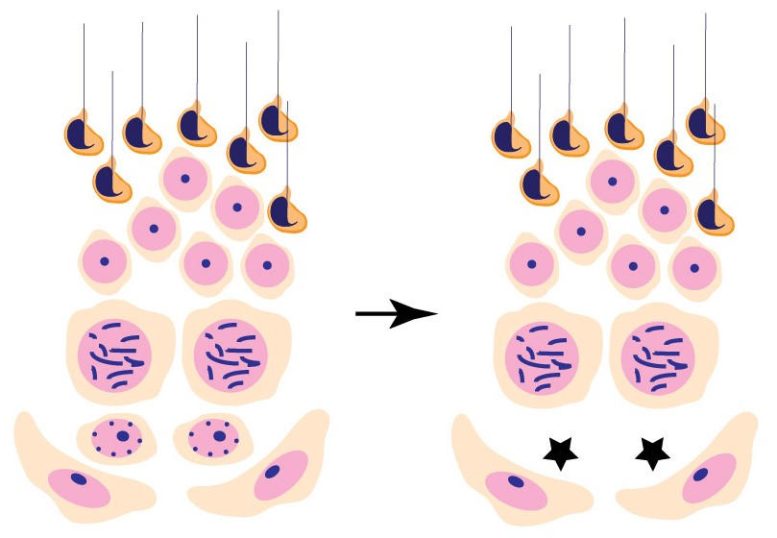
The Role of DOT1L in Sperm Production
The function of DOT1L in stem cell self-renewal was accidentally discovered by the researchers. The gene is widely expressed; mice with a mutant form of DOT1L in every cell do not survive beyond the embryonic stage. However, Wang and colleagues hypothesized that DOT1L could be involved in meiosis, the process of cell division that results in sperm and eggs, based on the genetic expression patterns of DOT1L. So they made the decision to investigate what would happen if they mutated the gene only in these germ cells.
“When we did this, the animals lived and appeared healthy,” Wang says. “When we looked closer, however, we found that the mice with the mutant DOT1L in their germ cells could complete an initial round of sperm production, but then the stem cells became exhausted and the mice lost all germ cells.”
This drop-off in sperm production could arise due to other problems. But various lines of evidence supported the link between DOT1L and a failure of stem cell self-renewal. In particular, the researchers found that the mice experienced a sequential loss of the various stages of sperm development, first failing to make spermatogonia and then spermatocytes, followed by round spermatids, and then elongated spermatids.
In a further experiment, the researchers observed what happened when DOT1L was inactivated in germ cells not from birth, but during adulthood. As soon as Wang and colleagues triggered the DOT1L loss, they observed the same sequential loss of sperm development they had seen in the mice born without DOT1L in their germ cells.
Previously, other scientific groups have studied DOT1L in the context of leukemia. Overexpression of the gene in the progenitors of blood cells can lead to malignancy. From that line of investigation, it was known that DOT1L acts as a histone methyltransferase, an enzyme that adds a methyl group to histones to influence gene expression.
DOT1L’s Mechanism: Histone Methylation and Gene Regulation
To see whether the same mechanism was responsible for the results Wang and his team had observed in sperm development, the researchers treated spermatogonial stem cells with a chemical that blocks the methyltransferase activity of DOT1L. When they did so, the stem cells’ ability to give rise to spermatogonia was significantly reduced. The treatment also impaired the ability of stem cells to tag histones with a methyl group. And when these treated stem cells were transplanted into otherwise healthy mice, the animals’ spermatogonial stem cell activity was cut in half.
The team found that DOT1L appeared to be regulating a gene family known as Hoxc, transcription factors that play significant roles in regulating the expression of a host of other genes.
“We think that DOT1L promotes the expression of these Hoxc genes by methylating them,” says Wang. “These transcription factors probably contribute to the stem cell self-renewal process. Finding out the details of that is a future direction for our work.”
A longer-term goal is to use factors like DOT1L and others involved in germline stem cell self-renewal to help people who have fertility challenges. The concept is to create germ cells from the ground up.
“That’s the future of this field: in vitro gametogenesis,” Wang says. “Reprogramming somatic cells to become spermatogonial stem cells is one of the steps. And then we’d have to figure out how to have those cells undergo meiosis. We’re in the early stages of envisioning how to accomplish this multi-step process, but identifying this self-renewal factor brings us one step closer.”
Reference: “Histone methyltransferase DOT1L is essential for self-renewal of germline stem cells” by Huijuan Lin, Keren Cheng, Hiroshi Kubota, Yemin Lan, Simone S. Riedel, Kazue Kakiuchi, Kotaro Sasaki, Kathrin M. Bernt, Marisa S. Bartolomei, Mengcheng Luo and P. Jeremy Wang, 23 June 2022, Genes & Development.
DOI: 10.1101/gad.349550.122
The study was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Natural Science Foundation of China, the China Scholarship Council, and the Japan Society for the Promotion of Science.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.