Urolithin A may enhance muscle endurance and mitochondrial health by promoting mitophagy, according to a new study.
Although not all measures of muscle function showed improvement, there were notable benefits in endurance and metabolism, particularly in older adults.
An oral supplement intended to stimulate a natural body process appears to promote muscle endurance and mitochondrial health in humans. New research suggests that the supplement, urolithin A, may help improve or prolong muscle activity in people who are aging or who have diseases that make exercise difficult.
The paper was published in JAMA Network Open.
Mitochondrial Health and Aging
“This is relevant both to people with chronic diseases and people who want to be more active later in life,” said the lead author, David Marcinek, a professor of radiology at the University of Washington School of Medicine. His research has focused on the role of mitochondria in aging and chronic disease.
Urolithin A is a byproduct of a person’s gut bacteria and a diet comprising polyphenols found in pomegranates, berries, and nuts. Because diet, age, genetics, and disease affect the makeup of the gut microbiome, people produce urolithin A at variable rates. The compound also is produced and sold by dietary supplement companies.
Research Findings on Urolithin A
Supplemental urolithin A has been shown in animal tests and molecular studies of humans to stimulate mitophagy, a process that Marcinek explained as “mitochondrial quality control.”
“Mitochondria are like batteries that power the cells in your body,” he said. “But over time they break down. The process of mitophagy recognizes this failure and proactively tears down the mitochondria, reducing it to elemental components that a cell can reuse. But with aging, mitophagy becomes less efficient and your body accumulates this pool of failing mitochondria. It’s one way that muscles become less functional as we age.”
The researchers studied a small cohort of people over age 65 who were randomized to receive a placebo or a daily supplement of 1,000 mg urolithin A for four months. Each of the 66 subjects was confirmed at the outset to have average or subpar capacity to produce adenosine triphosphate (ATP), which mitochondria produce to help cells perform myriad functions.
The investigators hypothesized that, if the urolithin A supplement indeed boosted mitophagy, the test cohort would experience improved muscle function and greater ATP output.
Study Outcomes and Potential Benefits
Across both cohorts, two comparisons of muscle function were found to support the thesis, but two others did not:
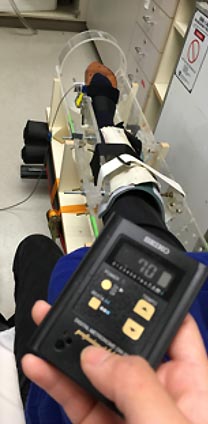
- Two measures of muscle endurance were improved in the supplemented group compared to the placebo group. Endurance was measured with exercises involving the hand (first dorsal interosseous, between thumb and forefinger) and leg (tibialis anterior, alongside the shinbone.) Researchers measured the increase in the number of muscle contractions until fatigue between a baseline test and the final test four months later.
- Measures of distance covered during a six-minute walk improved markedly between tests at baseline and four months in both the supplement and placebo groups. However, researchers saw no significant effect of the supplement compared with the placebo.
- Measures (via magnetic resonance spectroscopy) of improvement of maximal ATP production did not change significantly between baseline and four months in either group.
“Even though we did not observe an effect of the supplement in whole body function (via six-minute measure and ATP production),” Marcinek said, “these results are still exciting because they demonstrate that just taking a supplement for a short duration actually improved muscle endurance. Fatigue resistance got better in the absence of exercise.”

Plasma samples also were collected from study participants at the outset, at two months, and four months. The purpose was to assess the supplement’s potential effect on urolithin A bioavailability and on biomarkers of mitochondrial health and inflammation. In the test cohort, Urolithin A was associated with a significant reduction in several acylcarnitines and ceramides implicated for their roles in metabolic disorders involving mitochondria, the investigators reported.
“I think these changes suggest that the treatment affects the metabolic condition of people. Even though it didn’t affect the maximum ATP production, it improved test subjects’ general metabolism,” Marcinek said.
He added that urolithin A supplements could have potential to benefit people who cannot get the exercise they want due to poor muscle health or disease.
“Just getting them over that point where exercise is possible – a walk around the block or climbing some stairs – might help a person build their own health.”
The study’s funder, Amazentis, of Lausanne, Switzerland, manufactures the urolithin A supplement used in the trial.
Reference: “Effect of Urolithin A Supplementation on Muscle Endurance and Mitochondrial Health in Older Adults” by Sophia Liu, PhD; Davide D’Amico, PhD; Eric Shankland, PhD; Saakshi Bhayana, BS; Jose M. Garcia, MD, PhD; Patrick Aebischer, MD; Chris Rinsch, PhD; Anurag Singh, MD, PhD and David J. Marcinek, PhD, 20 January 2022, JAMA Network Open.
DOI: 10.1001/jamanetworkopen.2021.44279
Dr. Liu reported reported receiving grants from Amazentis SA during the conduct of the study as well as drugs at no cost for research use from Stealth BioTherapeutics and supplements at no cost for research use from AstaReal outside the submitted work. Dr. D’Amico reported being an employee of Amazentis SA during the conduct of the study and receiving personal fees from Amazentis SA outside the submitted work. Dr. Shankland reported receiving grants from Amazentis SA during the conduct of the study as well as drugs at no cost for research use from Stealth BioTherapeutics and supplements at no cost for research use from AstaReal outside the submitted work. Ms. Bhyana reported receiving grants from Amazentis SA during the conduct of the study as well as drugs at no cost for research use from Stealth BioTherapeutics and supplements at no cost for research use from AstaReal outside the submitted work. Dr. Garcia reported being a full-time employee of the US government during the conduct of the study. Dr. Aebischer reported being the chair of the board and a shareholder of Amazentis SA during the conduct of the study; holding a patent for US 11,020,373 issued, a patent for US 10,906,883 issued, a patent for US 20180256538 pending, and a patent for US 20180256539 pending. Dr. Singh reported being an employee of Amazentis SA during the conduct of the study and holding a patent for US 20180256538 pending and the patent for US 20180256539 pending. Dr. Marcinek reported receiving grants from Amazentis SA during the conduct of the study as well as drugs at no cost for research use from Stealth BioTherapeutics, grants from Boehringer Ingelheim, and supplements at no cost for research use from AstaReal outside the submitted work.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
This is one of those possible beneficial properties of CBD that has the greatest amount of evidence – after the benefits in the treatment of epilepsy, however, “there is still much less evidence here than in matters related to epilepsy,” says Bonn-Miller.
I have severe narcolepsy, depression and am intolerant to the extreme heat where I live. I can barely move. I’ve lost so much strength and indurance I got very out of breath riding my ebike half a block down hill. It would be life changing if something like this helped.