In a new study, researchers successfully engrafted insulin-producing cells into a diabetic mouse model, reversing diabetic symptoms in the animal in as little as 10 days.
Researchers have made a significant first step with newly engineered biomaterials for cell transplantation that could help lead to a possible cure for Type 1 diabetes, which affects about 3 million Americans.
Georgia Tech engineers and Emory University clinicians have successfully engrafted insulin-producing cells into a diabetic mouse model, reversing diabetic symptoms in the animal in as little as 10 days.
The research team engineered a biomaterial to protect the cluster of insulin-producing cells – donor pancreatic islets – during injection. The material also contains proteins to foster blood vessel formation that allow the cells to successfully graft, survive, and function within the body.
“It’s very promising,” said Andrés Garcia, Georgia Tech professor of mechanical engineering. “There is a lot of excitement because not only can we get the islets to survive and function, but we can also cure diabetes with fewer islets than are normally needed.”
The research article – a partnership with Emory’s Dr. Robert Taylor and Dr. Peter Thule that was funded in part by the JDRF, the leading global organization funding Type 1 diabetes research – is published in the June issue of the journal Biomaterials.
Organizations such as JDRF are dedicated to finding a cure for Type 1 diabetes, a chronic disease that occurs when the pancreas produces little or no insulin, a hormone that allows the transport of sugar and other nutrients into tissues where they are converted to energy needed for daily life.
Pancreatic islet transplantation re-emerged as a promising therapy in the late 1990s. Patients with diabetes typically find it difficult to comply with multiple daily insulin injections, which only partially improve long-term outcomes. Successful islet transplantation would remove the need for patients to administer insulin. While islet transplantation trials have had some success, and control of glucose levels is often improved, diabetic symptoms have returned in most patients and they have had to revert to using some insulin.
Unsuccessful transplants can be attributed to several factors, researchers say. The current technique of injecting islets directly into the blood vessels in the liver causes approximately half of the cells to die due to exposure to blood clotting reactions. Also, the islets – metabolically active cells that require significant blood flow – have problems hooking up to blood vessels once in the body and die off over time.
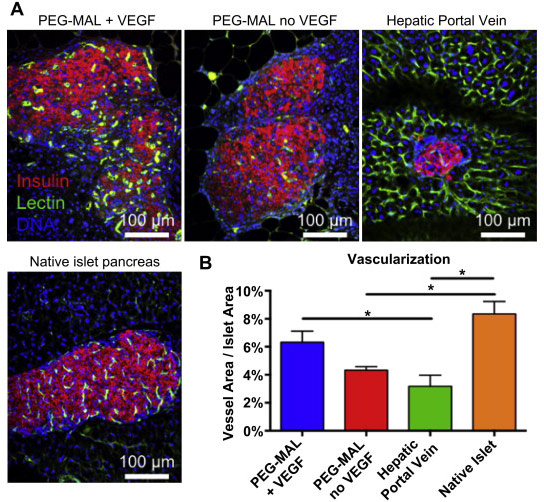
Georgia Tech and Emory researchers engineered a hydrogel, a material compatible with biological tissues that is a promising therapeutic delivery vehicle. This water-swollen, cross-linked polymer surrounds the insulin-producing cells and protects them during injection. The hydrogel containing the islets was delivered to a new injection site on the outside of the small intestine, thus avoiding direct injection into the blood stream.
Once in the body, the hydrogel degrades in a controlled fashion to release a growth factor protein that promotes blood vessel formation and connection of the transplanted islets to these new vessels. In the study, the blood vessels effectively grew into the biomaterial and successfully connected to the insulin-producing cells.
Four weeks after the transplantation, diabetic mice treated with the hydrogel had normal glucose levels, and the delivered islets were alive and vascularized to the same extent as islets in a healthy mouse pancreas. The technique also required fewer islets than previous transplantation attempts, which may allow doctors to treat more patients with limited donor samples. Currently, donor cells from two to three cadavers are needed for one patient.
While the new biomaterial and injection technique is promising, the study used genetically identical mice and therefore did not address immune rejection issues common to human applications. The research team has funding from JDRF to study whether an immune barrier they created will allow the cells to be accepted in genetically different mice models. If successful, the trials could move to larger animals.
“We broke up our strategy into two steps,” said Garcia, a member of Georgia Tech’s Petit Institute for Bioengineering and Bioscience. “We have shown that when delivered in the material we engineered, the islets will survive and graft. Now we must address immune acceptance issues.”
Most people with Type 1 diabetes currently manage their blood glucose levels with multiple daily insulin injections or by using an insulin pump. But insulin therapy has limitations. It requires careful measurement of blood glucose levels, accurate dosage calculations, and regular compliance to be effective.
Reference: “Vasculogenic bio-synthetic hydrogel for enhancement of pancreatic islet engraftment and function in type 1 diabetes” by Edward A. Phelps, Devon M. Headen, W. Robert Taylor, Peter M. Thulé and Andrés J. García, 26 March 2013, Biomaterials.
DOI: 10.1016/j.biomaterials.2013.03.012
This work was also funded by the Regenerative Engineering and Medicine Center at Georgia Tech and Emory, and the Atlanta Clinical and Translation Science Institute under PHS grant UL RR025008 from the Clinical and Translational Science Award Program.
The Center for Pediatric Healthcare Technology Innovation at Georgia Tech, the Department of Veterans Affairs Merit Review Program and the National Institutes of Health’s National Institute of Diabetes and Digestive and Kidney Diseases (Grant R01 DK076801-01) helped fund the project as well.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.