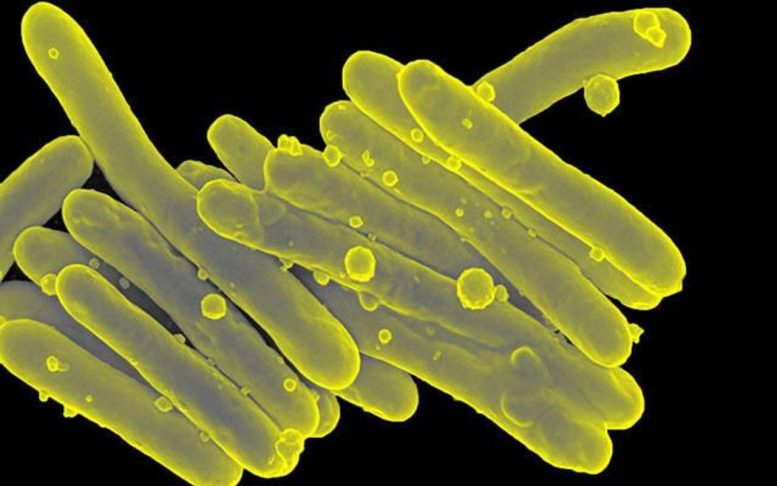
Twenty complex, antibiotic-resistant lung infections were treated by researchers using bacteria-eating viruses; there were no negative side effects, and more than half of the patients treated had favorable clinical results.
Researchers from the University of California San Diego School of Medicine and the University of Pittsburgh, leading an international team of researchers, present encouraging findings from the largest case series of patients treated with bacteriophage treatment for antibiotic-resistant infections.
Patients with cystic fibrosis or other chronic disorders that harm or destroy the lungs’ bronchi—the network of tubes that carry oxygen and other gases throughout the organs—are more likely to get non-tuberculosis Mycobacterium (NTM) infections.

The findings were recently published in the journal Clinical Infectious Diseases.
Non-tuberculosis Mycobacterium (NTM) infections are increasingly common among patients with cystic fibrosis or other chronic diseases that damage or destroy the lungs’ bronchi — the network of tubes that transport oxygen and other gases throughout the organs.
Due to bacterial resistance to antibiotics, which have long been the standard of care, treating NTM infections is challenging, especially those brought on by Mycobacterium abscessus. According to the Centers for Disease Control, 35,000 people die each year in the United States as a consequence of roughly 3 million infections that are resistant to all antibiotics.
Viruses known as bacteriophages have developed to specifically target and destroy certain bacterial species or strains. Wherever bacteria live, phages may be found in greater numbers than all other living forms combined. They were first identified in the early 20th century, and since then, as antibiotic-resistant bacteria have grown and spread, more and more research has been done on them to determine their medicinal potential.
In 2016, scientists and physicians at the UC San Diego School of Medicine and UC San Diego Health used an experimental intravenous phage therapy to successfully treat and cure colleague Tom Patterson, Ph.D., who was near death from a multidrug-resistant bacterial infection. Patterson’s was the first documented case in the U.S. to employ intravenous phages to eradicate a systemic bacterial infection. Subsequent successful cases helped lead to the creation of the Center for Innovative Phage Applications and Therapeutics (IPATH) at UC San Diego, the first such center in North America.
“We think this is a revolutionary topic and study that evolved from our original Tom Patterson case report,” said co-corresponding author Constance Benson, MD, professor of medicine and global public health at UC San Diego School of Medicine. “It promises to be highly cited as we at IPATH and others work on expanding the uses of phage therapy.”
Currently, those uses are limited, in part because each phage species seeks and destroys only one bacterial species and the current armamentarium of known therapeutically useful phages is relatively small. As a result, phage therapy testing is currently constrained to experimental treatments where all other viable alternatives are failing or have failed.
The new study involved a cohort of 20 patients with complex, antibiotic-refractory mycobacterial infections. All of the patients exhibited varying underlying conditions; most had cystic fibrosis (CF), an inherited, progressive disease that causes severe damage to the lungs and other organs. Currently, there is no cure for CF. The average lifespan for persons with CF who live to adulthood is approximately 44 years.
Participating patients in the study qualified under the U.S. Food and Drug Administration’s “compassionate use” provision, which allows testing of investigational drugs or products for life-threatening conditions when no comparable or satisfactory alternative therapies are available.
Benson, co-corresponding author Graham F. Hatfull, Ph.D., Eberly Family Professor of Biotechnology at the University of Pittsburgh, and colleagues screened 200 patients with symptomatic lung disease to identify bacterial strains likely to be susceptible to phages and identified 55 strains.
Phages were administered to the 20 study participants intravenously, by aerosolization through a nebulizer, or by using both methods twice daily over an average course of six months, though some patients had shorter or longer treatments based on clinical or microbiologic response.
Patients were monitored for adverse effects, signs of symptomatic improvement or reduced bacterial presence, the emergence of phage resistance, and/or neutralization of phages by the patients’ immune systems.
Results
The authors reported no adverse reactions to phage therapy in any of the patients, regardless of the type of bacterial infection, types of phages used, or method of treatment. Eleven of the 20 patients displayed some measure of symptom improvement or reduced bacterial presence. Five patients had inconclusive outcomes and four exhibited no response to treatment.
In eight patients, there was a noted increase in neutralizing antibodies, which may have contributed to the lack of treatment response in four cases. Eleven patients were treated with a single phage, with no indications of phage resistance observed.
“Given the complexity and great variation of these patients and their individual conditions, it is not possible to draw any broad conclusions, except that phage treatment of mycobacterial infections shows promise and should be explored further,” said Benson, “especially for treating patients with few or no other good options.”
Hatfull said the study provided several insights into how therapeutic phages might be effectively used.
First, he said, it underscored the need to expand significantly the repertoire of useful phages, whether developing them from isolated strains or creating synthetic versions, an emerging enterprise.
Second, the lack of phage resistance was encouraging, supporting the use of a single phage treatment, though where more than one suitable phage is available, the authors suggested cycling their administration to circumvent neutralization by the patient’s immune system.
Third, optimal administration of phages, whether intravenous or aerosolization, may depend upon the nature of the infection and whether the patient’s immune system is compromised.
Fourth, because phages appear to be well tolerated with no adverse effects, higher doses and longer periods of treatment might be possible and advisable.
“All of the limitations we observed and have documented are not insurmountable,” said Hatfull. “These case studies suggest that phage treatments may be valuable tools for clinical control of NTM infections.”
Co-author Robert Schooley, MD, professor of medicine and an infectious disease expert at UC San Diego School of Medicine who is co-director of IPATH and helped lead the clinical team that treated and cured Patterson in 2016, took a longer view:
“In phages, evolution has produced an effective killer of bacteria, one that offers enormous potential in the worldwide fight against antibiotic resistance. This paper is a glimpse of what might and can be. It starts with NTM infections, but the number of antibiotic-resistant bacterial species out there is large and growing. This is another step, an important one, in a fight that will likely never end.”
Reference: “Phage Therapy of Mycobacterium Infections: Compassionate-use of Phages in Twenty Patients with Drug-Resistant Mycobacterial Disease” by Rebekah M. Dedrick, Bailey E. Smith, Madison Cristinziano, Krista G. Freeman, Deborah Jacobs-Sera, Yvonne Belessis, A. Whitney Brown, Keira A. Cohen, Rebecca M. Davidson, David van Duin, Andrew Gainey, Cristina Berastegui Garcia, C.R. Robert George, Ghady Haidar, Winnie Ip, Jonathan Iredell, Ameneh Khatami, Jessica S. Little, Kirsi Malmivaara, Brendan J. McMullan, David E. Michalik, Andrea Moscatelli, Jerry A. Nick, Maria G. Tupayachi Ortiz, Hari M. Polenakovik, Paul D. Robinson, Mikael Skurnik, Daniel A. Solomon, James Soothill, Helen Spencer, Peter Wark, Austen Worth, Robert T. Schooley, Constance A. Benson and Graham F. Hatfull, 9 June 2022, Clinical Infectious Diseases.
DOI: 10.1093/cid/ciac453
The study was funded by the National Institutes of Health, the Howard Hughes Medical Institute, the Cystic Fibrosis Foundation, The Fowler Fund for Phage Research, the National Heart Lung and Brain Institute, the Burroughs Wellcome Fund, the Jane and Aatos Erkko Foundation, the Mallory Smith Legacy Fund, and the UC San Diego Chancellor’s Fund.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
I have the rare lung disease MAC and I am interested in this page there are no phage places I know of near me South Carolina now I was also diagnosed with stage 3 esophagus cancer Adenocarcinoma