Scientists in Germany have achieved something never seen before: creating a stable boron-carbon triple bond.
While triple bonds exist between many element pairs, boron and carbon had stubbornly resisted. This new discovery not only fills a long-standing gap in chemistry but also opens the door to new chemical reactions and potential innovations in synthesis. The unique instability of the bond makes it highly reactive, which could lead to exciting new materials and applications.
Uncovering the Power of Triple Bonds
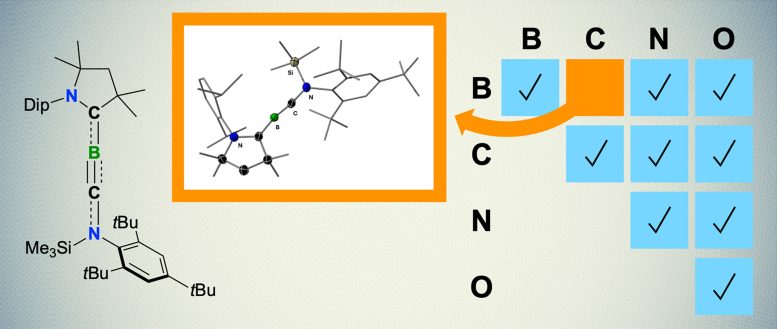
Boron, carbon, nitrogen, and oxygen are four elements capable of forming triple bonds due to their similar electronic properties. Common examples include carbon monoxide (CO), a gas composed of one carbon and one oxygen atom, and nitrogen gas (N2), which makes up most of Earth’s atmosphere and consists of two nitrogen atoms bonded by a triple bond.
Triple bonds are well-documented between all possible pairings of these four elements — except for boron and carbon. This gap is surprising, given that boron and carbon are known to form stable double bonds, and triple bonds between two carbon atoms or two boron atoms have long been established in chemistry.
A Breakthrough in Boron Chemistry
Now, chemists at Julius-Maximilians-Universität (JMU) Würzburg in Germany have filled this gap. Led by boron expert Professor Holger Braunschweig, the team has successfully synthesized the first-ever molecule containing a boron-carbon triple bond, known as a boryne. This groundbreaking compound exists as an orange solid at room temperature. The researchers have characterized the molecule and conducted initial studies on its reactivity, with their findings published on March 4 in Nature Synthesis.
Boron Atom in an Uncomfortable Situation
In the novel molecule, the boron atom is in a linear arrangement with carbon atoms. “In combination with the triple bond, this is about as uncomfortable as it gets for boron, requiring very special conditions,” says Dr. Rian Dewhurst, co-author of the study. This is why it has taken so long to synthesize such a triple bond for the first time.
What interests the Würzburg chemists about the new molecule: “Compounds in which individual atoms feel ‘uncomfortable’ often show a very interesting reactivity,” explains Maximilian Michel, the doctoral student who made the molecule in the laboratory.
It is precisely this reactivity that the team’s further work is now focusing on. Ultimately, this may result in innovative tools for chemical syntheses. The findings could also be helpful for a better understanding of chemical bonds and structures.
Inspiration for Other Researchers
“Another benefit that is often overlooked: Basic research like ours inspires other researchers to put their efforts and imagination into synthesizing compounds that might seem improbable,” says Rian Dewhurst. “World-changing advances often emerge from these kinds of crazy ideas.” Teflon, for example, was discovered during research originally aimed at developing new refrigerants, while the well-known product superglue emerged by chance during attempts to produce transparent plastics.
Reference: “The synthesis of a neutral boryne” by Maximilian Michel, Sourav Kar, Lukas Endres, Rian D. Dewhurst, Bernd Engels and Holger Braunschweig, 4 March 2025, Nature Synthesis.
DOI: 10.1038/s44160-025-00763-1
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Great Work Guys. 🤓