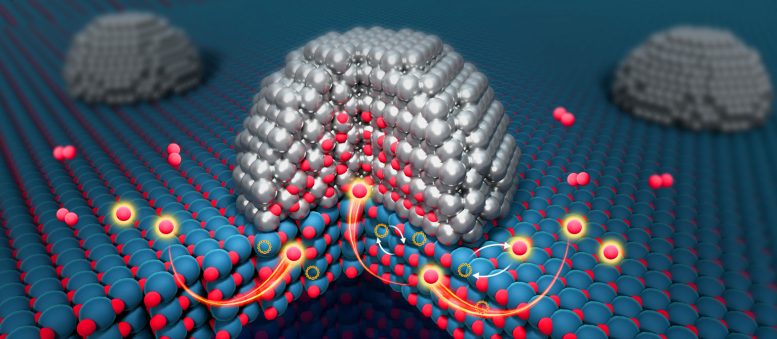
A new study reveals an unexpected pathway in catalytic systems, where oxygen does not remain confined to surfaces but can move through deeper regions of the material.
A team of scientists in China has uncovered a hidden pathway inside catalysts that could reshape how chemical reactions are designed and controlled. Led by Tao Zhang and Yanqiang Huang at the Dalian Institute of Chemical Physics, along with collaborators Wei Liu and Yanggang Wang, the researchers directly tracked how oxygen moves through catalytic materials. What they found challenges decades of assumptions.
Using advanced environmental transmission electron microscopy, the team captured the first real-time evidence of oxygen traveling through the interior of a catalyst, not just along its surface. Their results, published in Nature, suggest that large portions of catalytic materials previously considered inactive may actually play an important role.
Catalysts are essential to modern industry, speeding up reactions used in everything from fuel production to pollution control. A key process in many of these reactions is “spillover,” where atoms such as hydrogen or oxygen migrate between a metal and the material supporting it.
Until now, scientists believed this movement was largely confined to the outer surface. That assumption shaped how catalysts have been designed for decades, with most efforts focused on maximizing surface area.
Now, the researchers show that oxygen atoms can move through the bulk of the material itself, opening up an entirely new region for chemical activity.
Visualizing Oxygen Movement
To observe this effect, the team used titanium dioxide (TiO2), a widely used material known for its ability to store and release oxygen. It is already common in applications such as photocatalysis, environmental cleanup, and solar energy research.
By pairing TiO2 with ruthenium, a metal often used in catalytic systems, the researchers created a model system that allowed them to watch oxygen atoms in motion at the atomic scale.
Instead of remaining on the surface, oxygen atoms were seen moving from layers buried three to five atoms deep within the TiO2 toward the metal interface. This movement is driven by differences in oxygen chemical potential, essentially a gradient that pulls oxygen atoms through the material.
The study also revealed a surprising level of control at the boundary between the metal and the support material.
“A channel has been disclosed in TiO2 support to facilitate oxygen spillover, meanwhile the metal-support interface acts like an atomic-scale guard, controlling whether oxygen spillover can pass through. This finding inspires a new strategy for utilizing catalyst bulk that is conventionally believed useless in catalysis,” said Wei Liu.
Implications for Catalyst Design
“This unique oxygen spillover in our work enables the bulk of a catalyst, which is otherwise inaccessible to reactants, to contribute to mass transfer during catalytic reactions, underscoring the critical importance of interface engineering in controlling spillover behavior,” said Prof. Yanqiang Huang.
The findings build on decades of research into metal-support interactions. About 50 years ago, scientists discovered that metal particles can become covered by oxide supports such as TiO2 under strongly reducing conditions, reducing their ability to adsorb molecules like H2 and CO. Traditionally, these interactions were thought to involve only the outer surfaces where the metal and support meet.
Extending the Metal–Support Interaction Concept
This study expands that view by showing that oxygen spillover can also occur through internal interfaces within the catalyst. These regions, which are normally inaccessible to reacting molecules, can still play a role in moving material during reactions.
The results highlight the importance of carefully designing interfaces in catalysts and demonstrate how advanced imaging techniques can reveal reaction pathways at the level of individual particles.
Looking ahead, the team plans to continue developing this approach. “Taking this excellent opportunity, we can improve architecture of catalysis from two-dimensional surface reactions to the three dimensional ‘surface–interface–bulk’ synergy. It provides fresh insights into interfacial atomic engineering in heterogeneous catalysis and the dynamic catalytic behavior of supported metal catalyst. The next goal is to develop practical catalysts that utilize the bulk to directly contribute to chemical reactions,” said Tao Zhang.
Reference: “Imaging interface-controlled bulk oxygen spillover” by Weijue Wang, Hongbin Xu, Shuhui Liu, Xiaofeng Yang, Wei Liu, Yang-Gang Wang, Yanqiang Huang and Tao Zhang, 15 April 2026, Nature.
DOI: 10.1038/s41586-026-10324-x
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.