
Researchers uncover an unexpected route to better lithium-sulfur batteries by visualizing reactions at the atomic scale.
The journey from a laboratory discovery to real-world application can be extensive and fraught with challenges. Take the lithium-sulfur battery, for instance. While it boasts significant benefits over the existing lithium-ion batteries used in vehicles, it still hasn’t made a substantial mark on the market despite years of rigorous development.
That situation could change in the future thanks to the efforts of scientists at the U.S. Department of Energy’s (DOE) Argonne National Laboratory. Over the past decade, they have made several pivotal discoveries related to lithium-sulfur batteries. Their latest revelation, published in Nature, unlocks a previously unknown reaction mechanism that addresses a major shortcoming — the batteries’ very short lifetimes.
Gui-Liang Xu, a chemist in Argonne’s Chemical Sciences and Engineering division, stated “Our team’s efforts could bring the U.S. one large step closer to a greener and more sustainable transportation landscape.”
Advantages Over Lithium-Ion Batteries
Lithium-sulfur batteries offer three significant advantages over current lithium-ion batteries. Firstly, they can store two to three times more energy in a given volume, resulting in longer vehicle ranges. Secondly, their lower cost, facilitated by the abundance and affordability of sulfur, makes them economically viable. Lastly, these batteries do not rely on critical resources like cobalt and nickel, which may face shortages in the future.
Despite these benefits, transitioning from laboratory success to commercial viability has proven elusive. Laboratory cells have shown promising results, but when scaled up to commercial size, their performance rapidly declines with repeated charge and discharge.
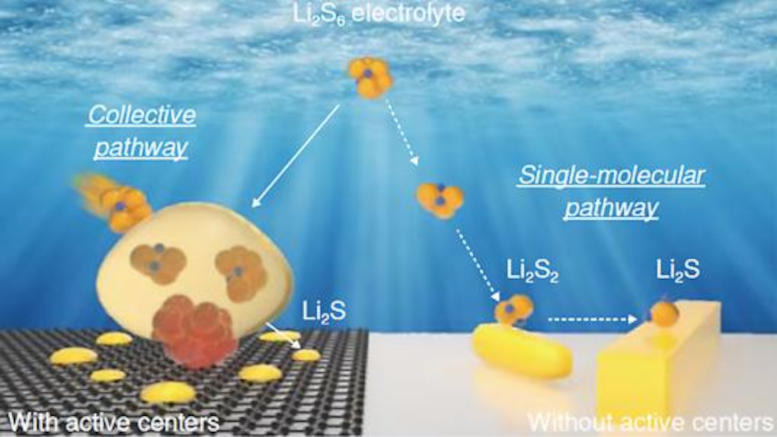
The underlying cause of this performance decline lies in the dissolution of sulfur from the cathode during discharge, leading to the formation of soluble lithium polysulfides (Li2S6). These compounds flow into the lithium metal negative electrode (anode) during charging, further exacerbating the issue. Consequently, the loss of sulfur from the cathode and alterations in the anode composition significantly hinder the battery’s performance during cycling.
A Breakthrough Catalyst to Prevent Sulfur Loss
In a recent earlier study, Argonne scientists developed a catalytic material that, when added in a small amount to the sulfur cathode, essentially eliminated the sulfur loss problem. While this catalyst showed promise in both laboratory and commercial-size cells, its atomic-scale working mechanism remained an enigma until now.
The team’s most recent research shed light on this mechanism. In the absence of the catalyst, lithium polysulfides form at the cathode surface and undergo a series of reactions, ultimately converting the cathode to lithium sulfide (Li2S).
“But the presence of a small amount of catalyst in the cathode makes all the difference,” Xu said. “A much different reaction pathway follows, one free of intermediate reaction steps.”
Key is the formation of dense nanoscale bubbles of lithium polysulfides on the cathode surface, which do not appear without the catalyst. These lithium polysulfides rapidly spread throughout the cathode structure during discharge and transform to lithium sulfide consisting of nanoscale crystallites. This process prevents the sulfur loss and performance decline in commercial-size cells.
In unlocking this black box around the reaction mechanism, the scientists employed cutting-edge characterization techniques. Analyses of the catalyst’s structure with the intense synchrotron X-ray beams at beamline 20-BM of the Advanced Photon Source, a DOE Office of Science user facility, revealed that it plays a critical role in the reaction pathway. The catalyst structure affects the shape and composition of the final product upon discharge, as well as the intermediate products. With the catalyst, nanocrystalline lithium sulfide forms upon full discharge. Without the catalyst, microscale rod-shaped structures form instead.
Visualizing Reactions at the Nanoscale
Another vital technique, developed at Xiamen University, allowed the team to visualize the electrode-electrolyte interface at the nanoscale while a test cell was working. This newly invented technique helped connect changes at the nanoscale to the behavior of an operating cell.
“Based on our exciting discovery, we will be doing more research to design even better sulfur cathodes,” Xu noted. “It would also be worthwhile to explore whether this mechanism applies to other next-generation batteries, such as sodium-sulfur.”
With this the team’s latest breakthrough, the future of lithium-sulfur batteries appears brighter, offering a more sustainable and eco-friendly solution for the transportation industry.
Reference: “Visualizing interfacial collective reaction behaviour of Li–S batteries” by Shiyuan Zhou, Jie Shi, Sangui Liu, Gen Li, Fei Pei, Youhu Chen, Junxian Deng, Qizheng Zheng, Jiayi Li, Chen Zhao, Inhui Hwang, Cheng-Jun Sun, Yuzi Liu, Yu Deng, Ling Huang, Yu Qiao, Gui-Liang Xu, Jian-Feng Chen, Khalil Amine, Shi-Gang Sun and Hong-Gang Liao, 6 September 2023, Nature.
DOI: 10.1038/s41586-023-06326-8
In addition to Xu, authors include Shiyuan Zhou, Jie Shi, Sangui Liu, Gen Li, Fei Pei, Youhu Chen, Junxian Deng, Qizheng Zheng, Jiayi Li, Chen Zhao, Inhui Hwang, Cheng-Jun Sun, Yuzi Liu, Yu Deng, Ling Huang, Yu Qiao, Jian-Feng Chen, Khalil Amine, Shi-Gang Sun and Hong-Gang Liao.
Other participating institutions include Xiamen University, Beijing University of Chemical Technology, and Nanjing University. The Argonne research was supported by the DOE Office of Vehicle Technologies in the Office of Energy Efficiency and Renewable Energy.
This research used resources from the Advanced Photon Source, a U.S. DOE Office of Science User Facility operated for the DOE Office of Science by Argonne National Laboratory under Contract No. DE-AC02-06CH11357.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.