A sweeping genetic screen reveals how the brain is built and exposes a hidden gene behind a newly discovered childhood brain disorder.
Which genes guide embryonic stem cells as they transform into brain cells, and what happens when that process breaks down? A new study published today (January 5) in Nature Neuroscience takes a major step toward answering those questions. The research was led by Prof. Sagiv Shifman from The Institute of Life Sciences at The Hebrew University of Jerusalem, working with Prof. Binnaz Yalcin from INSERM, France. Together, the team used large-scale CRISPR gene knockout experiments to pinpoint the genes that are essential for early brain development.
Mapping the Genes That Build the Brain
The goal of the study was simple but ambitious: to determine which genes are necessary for brain cells to develop correctly.
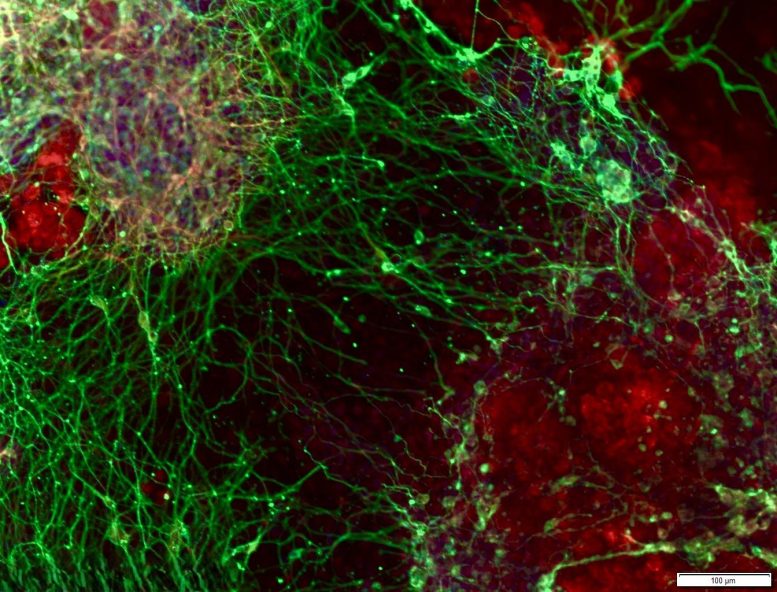
To do this, the researchers used CRISPR gene-editing tools to systematically shut down nearly 20,000 individual genes. These experiments were carried out in embryonic stem cells as they transitioned into brain cells. By disabling genes one at a time, the scientists could observe which ones were critical for the cells to successfully become neurons.
This approach allowed the team to chart the major stages of neural differentiation and identify 331 genes that are required for neuron formation. Many of these genes had never before been linked to early brain development. The findings help shed light on the genetic roots of neurodevelopmental conditions, including changes in brain size, autism, and developmental delay.
A New Genetic Cause of a Brain Development Disorder
One of the most important discoveries in the study was the identification of a gene called PEDS1 as the cause of a previously unrecognized neurodevelopmental disorder.
PEDS1 is necessary for producing plasmalogens, a specialized type of membrane phospholipid that is especially abundant in myelin, the fatty layer that insulates nerve fibers. The CRISPR screen showed that PEDS1 also plays a key role in forming nerve cells, and that losing this gene leads to a smaller brain. Based on these findings, the researchers suspected that PEDS1 defects could interfere with brain development in people as well.
That suspicion was confirmed through genetic testing of two unrelated families. In both cases, children with severe developmental symptoms were found to carry a rare mutation in PEDS1. The affected children showed developmental delay along with reduced brain size.
Confirming PEDS1’s Role in Brain Formation
To directly test whether PEDS1 loss causes these effects, the researchers disabled the gene in experimental models. The results confirmed that PEDS1 is required for normal brain development. Without it, nerve cells fail to form and migrate properly, which helps explain the symptoms seen in the children with the mutation.
Prof. Sagiv Shifman of the Faculty of Mathematics and Natural Sciences at Hebrew University explains: “By tracking the differentiation of embryonic stem cells into neural cells and systematically disrupting nearly all genes in the genome, we created a map of the genes essential for brain development. This map can help us better understand how the brain develops and identify genes linked to neurodevelopmental disorders that have yet to be discovered. Identifying PEDS1 as a genetic cause of developmental impairment in children, and clarifying its function, opens the door to improved diagnosis and genetic counseling for families, and may eventually support the development of targeted treatments.”
How Gene Pathways Shape Inheritance Patterns
The study also uncovered broader patterns that may help explain how different brain disorders are inherited. Genes involved in controlling other genes, such as those that regulate transcription and chromatin, are often linked to dominant disorders. In these cases, a mutation in just one copy of the gene can be enough to cause disease.
In contrast, disorders connected to metabolic genes, including PEDS1, tend to be recessive. This means both copies of the gene must be altered, usually with each parent carrying one mutated copy. Understanding this link between biological function and inheritance may help researchers and clinicians better identify and prioritize candidate disease genes.
Insights Into Autism and Developmental Delay
The researchers also created an “essentiality map” that shows when specific genes are required during development. This map helped clarify differences between autism and developmental delay at a biological level.
Genes that are essential across many stages of development were more strongly linked to developmental delay. Meanwhile, genes that are especially important during the formation of nerve cells were more closely associated with autism. These findings help explain why different genetic disruptions can produce overlapping symptoms and reinforce the idea that changes during early brain development play a role in autism.
Open Data to Support Future Research
The work was supported by the Israel Science Foundation (ISF), the ISF–Broad Institute Program, and the MAVRI Biomedical Research Program.
To make the findings widely available, the team launched an open online database containing the study’s results, allowing researchers around the world to explore the data.
Prof. Shifman added: “This was an excellent idea from PhD student Alana Amelan, who carried out a large part of the study and also created the website. We wanted our findings to serve the entire scientific community, supporting ongoing work on the genes we identified and helping researchers pinpoint additional genes involved in neurodevelopmental disorders.”
A Roadmap for Brain Development Research
Overall, the study delivers one of the most comprehensive genetic maps to date of early nervous system development. It also highlights the molecular basis of a newly identified disorder that disrupts brain growth.
These discoveries may improve genetic diagnosis for families affected by neurodevelopmental conditions and help lay the foundation for future research into prevention and treatment strategies.
Reference: “CRISPR knockout screens reveal genes and pathways essential for neuronal differentiation and implicate PEDS1 in neurodevelopment” by Alana Amelan, Stephan C. Collins, Nadirah S. Damseh, Nanako Hamada, Ahd Salim, Elad Dvir, Galya Monderer-Rothkoff, Tamar Harel, Koh-ichi Nagata, Binnaz Yalcin and Sagiv Shifman, 5 January 2026, Nature Neuroscience.
DOI: 10.1038/s41593-025-02165-0
Researchers: Alana Amelan, Stephan C. Collins, Nadirah S. Damseh, Nanako Hamada, Ahd Salim, Elad Dvir, Galya Monderer-Rothkoff, Tamar Harel, Koh-ichi Nagata, Binnaz Yalcin, and Sagiv Shifman.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
thanks for this
How
And w
Ho do i conaboit ths kind of research and be part of stidy if needed..