
Researchers discovered that a longevity gene from centenarians can reverse heart damage linked to progeria, suggesting a new approach to treating rapid and age-related heart aging.
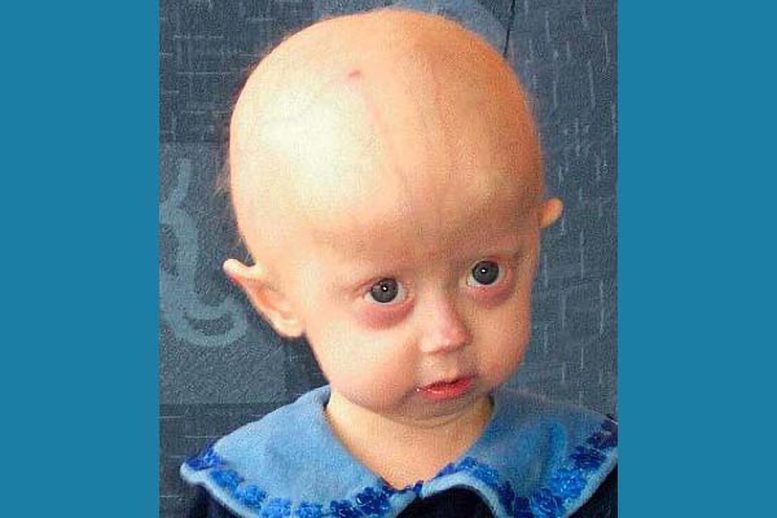
A major advancement has been made in understanding a rare genetic disorder that causes children to age prematurely. Scientists from the University of Bristol and IRCCS MultiMedica identified “longevity genes” found in people who live beyond 100 years, which appear to protect the heart and blood vessels during aging. Their study suggests these genes could potentially reverse the damage caused by this fatal condition.
Understanding progeria and its effects
Published in Signal Transduction and Targeted Therapy, this marks the first study to demonstrate that a gene from exceptionally long-lived individuals can slow cardiac aging in a model of progeria. Also known as Hutchinson-Gilford Progeria Syndrome (HGPS), progeria is a rare and deadly genetic disorder that causes children to experience “rapid-aging.”
HGPS results from a mutation in the LMNA gene, which produces a harmful protein called progerin. Most patients die in adolescence from heart-related complications, though a few—such as Sammy Basso, the longest-living known individual with progeria—survive longer. Sadly, Sammy passed away on 24 October last year at the age of 28.
Progerin harms cells by altering the structure of their nucleus, the “control center” of the cell, leading to premature aging signs, especially in cardiovascular tissue.
At present, the only treatment approved by the United States Food and Drug Administration (FDA) is lonafarnib, a drug that reduces the accumulation of progerin. A current clinical trial is investigating whether combining lonafarnib with another drug, Progerinin, may enhance its effectiveness.
Testing longevity genes as a new therapy
In the new study, Dr. Yan Qiu and Professor Paolo Madeddu from the Bristol Heart Institute, together with Professor Annibale Puca’s team at IRCCS MultiMedica in Italy, investigated whether genes from supercentenarians could protect children with progeria from progerin’s damaging effects.
The team focused on a ‘longevity gene’ found in centenarians, called LAV-BPIFB4. Previous research has shown that this gene helps keep the heart and blood vessels healthy during aging.
Using animal mice models genetically engineered to have Progeria, the research team was able to show early heart problems like those seen in children with the disease. The team found that a single injection of the longevity gene helped improve heart function, specifically how the heart relaxes and fills with blood – called diastolic function.
It reduced heart tissue damage – called fibrosis – and decreased the number of ‘aged’ cells in the heart. The gene also boosted the growth of new small blood vessels, which could help keep heart tissue healthy.
Gene therapy benefits in human cells
The team then tested the effect of the longevity gene in human cells from Progeria patients. Their findings showed adding the longevity gene to these cells reduced signs of aging and fibrosis, without changing progerin levels directly. This suggests the gene helps protect cells from the effects of progerin, rather than removing it. Importantly, the treatment doesn’t try to eliminate progerin but instead helps the body cope with its toxic effects.
New hope for heart protection and aging research
Dr Yan Qiu, Honorary Research Fellow in the Bristol Heart Institute at the University of Bristol, said: “Our research has identified a protective effect of a “supercentenarian longevity gene” against progeria heart dysfunction in both animal and cell models.
“The results offer hope to a new type of therapy for Progeria; one based on the natural biology of healthy aging rather than blocking the faulty protein. This approach, in time, could also help fight normal age-related heart disease.
“Our research brings new hope in the fight against Progeria and suggests the genetics of supercentenarians could lead to new treatments for premature or accelerated cardiac aging, which might help us all live longer, healthier lives.”
Toward future therapies and clinical applications
Professor Annibale Puca, Research Group Leader at IRCCS MultiMedica and Dean of the Faculty of Medicine at the University of Salerno, added: “This is the first study to indicate that a longevity-associated gene can counteract the cardiovascular damage caused by progeria.
“The results pave the way for new treatment strategies for this rare disease, which urgently requires innovative cardiovascular drugs capable of improving both long-term survival and patient quality of life. Looking ahead, the administration of the LAV-BPIFB4 gene through gene therapy could be replaced and/or complemented by new protein- or RNA-based delivery methods.
“We are currently conducting numerous studies to investigate the potential of LAV-BPIFB4 in counteracting the deterioration of the cardiovascular and immune systems in various pathological conditions, with the goal of translating these experimental findings into a new biologic drug.”
Reference: “A longevity-associated variant of the human BPIFB4 gene prevents diastolic dysfunction in progeria mice” by Yan Qiu, Monica Cattaneo, Anna Maciag, Annibale A. Puca and Paolo Madeddu, 29 September 2025, Signal Transduction and Targeted Therapy.
DOI: 10.1038/s41392-025-02416-3
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
2 Comments
studycan fing the DNA of the spine could benefit slower aging. the didion of the DNA is in the spine which can favor the whole body as long as its not overused
To add, gene therepy to modify thet DNA can be studied using properties of the skin of the apple. You would probably have to use an alloy to replenish the white blood cells.. maybe ginger