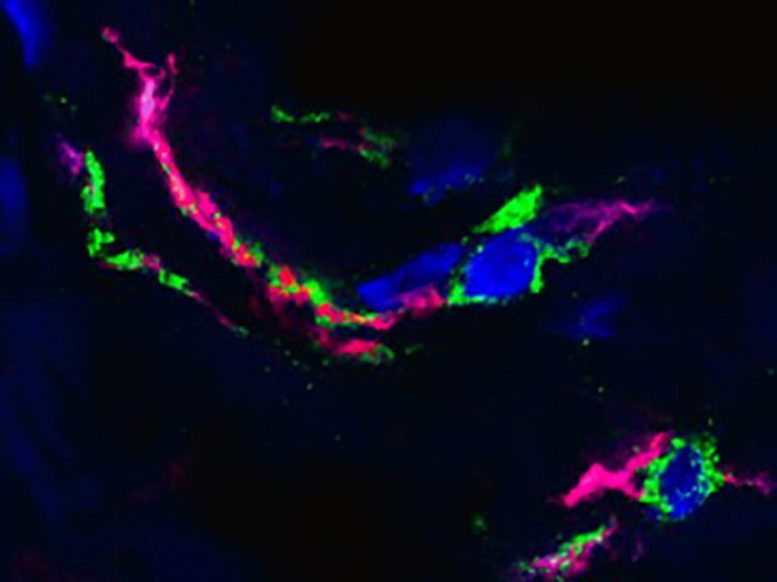
Sticky webs of DNA released from immune cells known as neutrophils may cause much of the tissue damage associated with severe COVID-19 infections, according to two new studies published on September 14, 2020, in the Journal of Experimental Medicine (JEM). The research, conducted by independent groups in Belgium and Brazil, suggests that blocking the release of these DNA webs could be a new therapeutic target for the management of severe forms of COVID-19.
While many people infected with the SARS-CoV-2 virus experience relatively mild symptoms, some patients mount an excessive inflammatory response that can damage the lungs and cause acute respiratory distress syndrome (ARDS), leading to low blood oxygen levels and, potentially, patient death. An early indicator of severe COVID-19 is an increased number of circulating neutrophils, a type of white blood cell. Neutrophils can catch and kill invading microbes by unwinding their DNA and extruding it from the cell to form sticky webs known as neutrophil extracellular traps (NETs). NETs can also damage surrounding tissue, however, and could therefore cause some of the lung pathology associated with severe COVID-19.
In one of the new studies, a research team from Liege University’s GIGA Institute led by Thomas Marichal, Cécile Oury, and Philippe Delvenne examined the lungs of patients who had succumbed to COVID-19 and found large numbers of NETs dispersed throughout the organ. The researchers saw many NETs in the airway compartment, where they often appeared to almost completely obstruct the small bronchioles and alveoli that mediate gas exchange. NETs were also formed at sites of inflammation located in the interstitial compartment between the alveoli and blood vessels, and could even be seen in the blood vessels themselves near tiny blood clots known as micro-thrombi that can restrict blood flow through the lungs and are a common pathological feature of severe COVID-19 patients.
“NETs can form a platform for the adhesion of platelets and other blood-clotting factors, but whether NETs actually contribute to the formation of COVID-19-associated pulmonary micro-thrombi will require further investigation,” says Thomas Marichal. “Altogether, our study supports the idea that targeting NETs in COVID-19 patients may help the clinical management of severe forms of COVID-19 by alleviating thrombotic events, excessive tissue-damaging inflammation, fibrosis, and airway obstruction.”
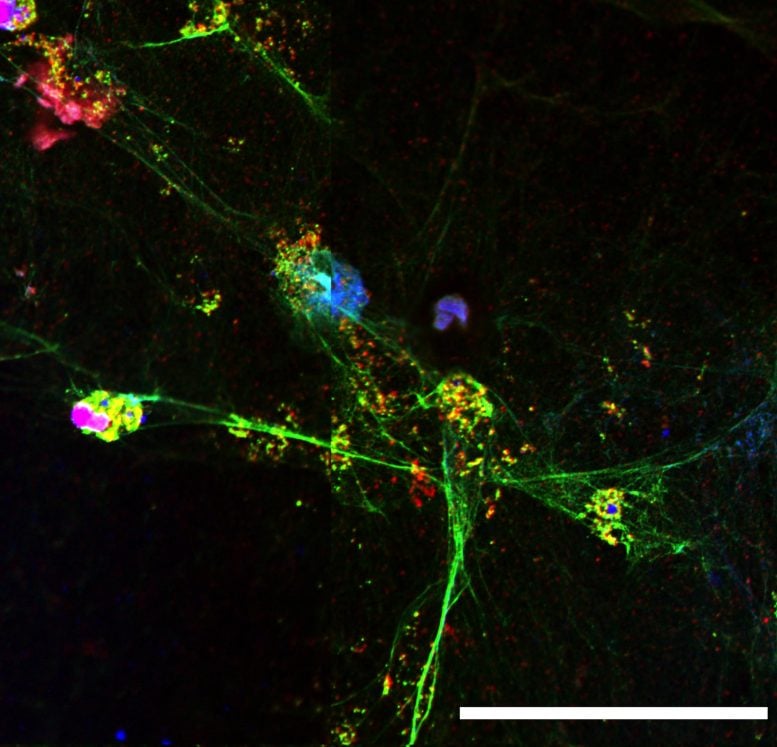
In the second study, a team of researchers led by Fernando Queiroz Cunha, Flavio Protasio Veras, and Thiago Mattar Cunha at the University of São Paulo also identified increased numbers of NETs in the lungs of severe COVID-19 patients and found that NET formation was elevated in COVID-19 patients’ blood plasma as well. Moreover, the researchers determined that SARS-CoV-2 can trigger the release of NETs by infecting neutrophils and replicating inside of them. NETs released from SARS-CoV-2-infected neutrophils induce the death of lung cells grown in the lab, the researchers found, but cell death is prevented if NET release is inhibited or the NETs are degraded by an enzyme that chews up DNA.
“Our study supports the use of inhibitors of NET synthesis or promoters of NET fragmentation as a strategy to ameliorate the organ damage associated with severe COVID-19,” says Fernando Queiroz Cunha.
References:
“Neutrophil extracellular traps infiltrate the lung airway, interstitial, and vascular compartments in severe COVID-19” by Coraline Radermecker, Nancy Detrembleur, Julien Guiot, Etienne Cavalier, Monique Henket, Céline d’Emal, Céline Vanwinge, Didier Cataldo, Cécile Oury, Philippe Delvenne and Thomas Marichal, 14 September 2020, Journal of Experimental Medicine.
DOI: 10.1084/jem.20201012
“SARS-CoV-2–triggered neutrophil extracellular traps mediate COVID-19 pathology” by Flavio Protasio Veras, Marjorie Cornejo Pontelli, Camila Meirelles Silva, Juliana E. Toller-Kawahisa, Mikhael de Lima, Daniele Carvalho Nascimento, Ayda Henriques Schneider, Diego Caetité, Lucas Alves Tavares, Isadora M. Paiva, Roberta Rosales, David Colón, Ronaldo Martins, Italo Araujo Castro, Glaucia M. Almeida, Maria Isabel Fernandes Lopes, Maíra Nilson Benatti, Letícia Pastorelli Bonjorno, Marcela Cavichioli Giannini, Rodrigo Luppino-Assad, Sérgio Luna Almeida, Fernando Vilar, Rodrigo Santana, Valdes R. Bollela, Maria Auxiliadora-Martins, Marcos Borges, Carlos Henrique Miranda, Antônio Pazin-Filho, Luis Lamberti P. da Silva, Larissa Cunha, Dario S. Zamboni, Felipe Dal-Pizzol, Luiz O. Leiria, Li Siyuan, Sabrina Batah, Alexandre Fabro, Thais Mauad, Marisa Dolhnikoff, Amaro Duarte-Neto, Paulo Saldiva, Thiago Mattar Cunha, José Carlos Alves-Filho, Eurico Arruda, Paulo Louzada-Junior, Renê Donizeti Oliveira, and Fernando Queiroz Cunha, 14 September 2020, Journal of Experimental Medicine.
DOI: 10.1084/jem.20201129
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.