Inside malaria parasites, tiny iron crystals spin like rockets, revealing a hidden weakness scientists may be able to exploit.
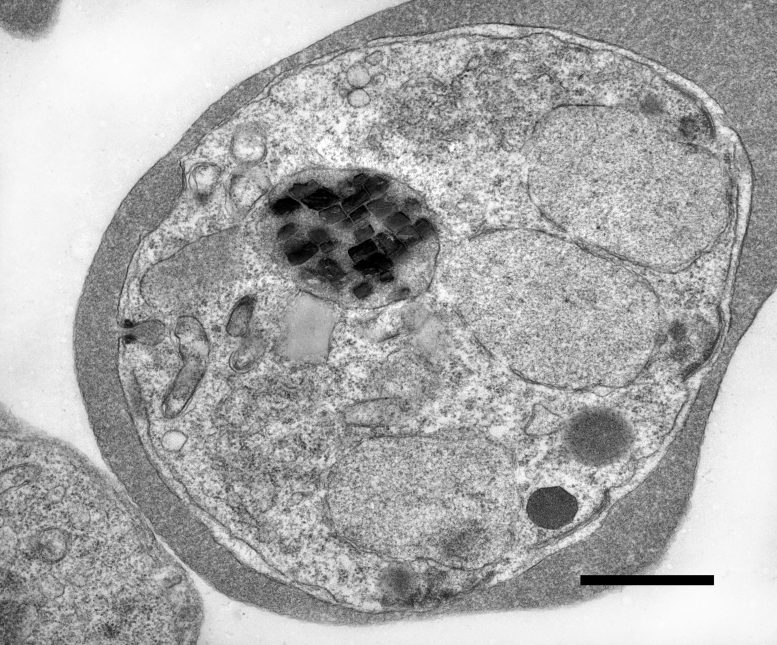
Inside every cell of the malaria-causing parasite Plasmodium falciparum sits a small compartment packed with microscopic iron crystals. While the parasite is alive, these crystals are constantly in motion. They spin, collide, and rebound inside their enclosed space, moving so rapidly and unpredictably that conventional scientific tools have struggled to measure them. When the parasite dies, the movement abruptly ends.
For years, these iron crystals have been a key target of antimalarial drugs, yet the reason behind their motion has remained unclear.
“People don’t talk about what they don’t understand, and because the motion of these crystals is so mysterious and bizarre, it’s been a blind spot for parasitology for decades,” says Paul Sigala, PhD, associate professor of biochemistry in the Spencer Fox Eccles School of Medicine (SFESOM) at the University of Utah.
Sigala and his colleagues have now uncovered the source of this movement. The crystals are powered by the same type of chemical reaction used to propel rockets into space.
The discovery may point to new ways of disrupting the malaria parasite and could also inform future efforts to design tiny machines that operate at the nanoscale. The results were published in the journal PNAS.
Biological rocket fuel
The researchers found that the crystals, made from an iron-containing compound called heme, move by driving a chemical reaction that breaks hydrogen peroxide down into water and oxygen. This reaction releases energy, providing the force that sets the crystals in motion and keeps them spinning.
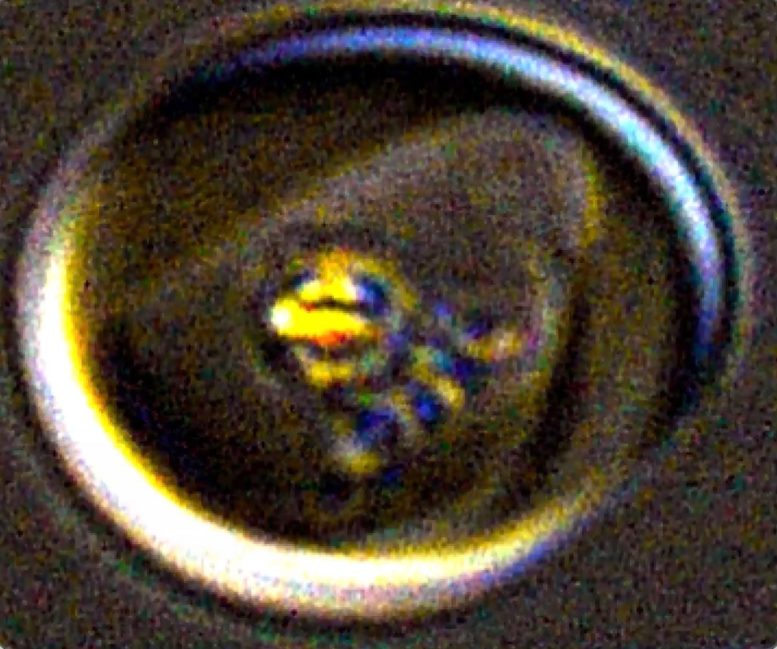
Microscope video of iron crystals spinning in a live malaria parasite (guitar pick shape) within a human red blood cell (larger circle). Credit: Erica Hastings
It’s a form of propulsion common in aerospace engineering, where peroxide fuel launches satellites into orbit, but previously unknown in biology. “This hydrogen peroxide decomposition has been used to power large-scale rockets,” says Erica Hastings, PhD, postdoctoral fellow in biochemistry in the SFESOM. “But I don’t think it has ever been observed in biological systems.”
Hydrogen peroxide is found at high levels inside the microscopic compartment that contains iron crystals, and parasites make the compound as a waste product, so it had stood out to the researchers as a potential chemical fuel that might power the crystals’ motion. Indeed, the scientists found that hydrogen peroxide on its own was enough to set purified crystals spinning—no parasite required.
Conversely, when the researchers raised malaria parasites at unusually low levels of oxygen, which lowers the amount of peroxide parasites produce, the crystals decelerated to about half their normal speed, even though the parasites were otherwise healthy.

Crystal motion may aid parasite survival
The researchers suspect that the frenetic motion of the crystals may be important for malaria parasites to stay alive, and they have a few ideas why. Peroxide itself is extremely toxic to cells. The spinning crystals might be a way for the malaria organism to “burn off” excess toxic peroxide before it can cause harmful chemical reactions and damage the parasite.
Sigala adds that the spinning motion might also help the parasite quickly deal with excess heme by keeping crystals from clumping together. Clumped-up crystals would prevent the parasite from storing additional heme as quickly, because they’d have less available surface to add new heme to. By keeping the crystals in constant motion, the malaria parasite may ensure that it’s able to sequester additional heme efficiently.
Powering new robots and new drugs
The spinning crystals are the first known example in biology of a self-propelled metallic nanoparticle, the researchers say. But they suspect that this phenomenon is much more widespread.
The new findings could inspire improved designs for microscopic robots, the researchers add.
“Nano-engineered self-propelling particles can be used for a variety of industrial and drug delivery applications, and we think there are potential insights that will come from these results,” Sigala says.

The results could also eventually lead to better antimalarial drugs, the researchers say. “We think that the breakdown of hydrogen peroxide likely makes an important contribution to reducing cellular stress,” Sigala says. “If there are ways to block the chemistry at the crystal surface, that alone might be sufficient to kill parasites.”
Their tiny chemical rockets are wildly different from any known aspect of human biology—and that means that they’re a powerful potential drug target. Drugs that target such a parasite-unique mechanism are much less likely to have dangerous side effects. “If we target a drug to an area that’s very different from human cells, then it’s probably not going to have extreme side effects,” Hastings explains. “If we can define how this parasite is different from our bodies, it gives us access to new directions for medications.”
Reference: “Chemical propulsion of hemozoin crystal motion in malaria parasites” by Erica M. Hastings, Tomasz Skóra, Keith R. Carney, Henry C. Fu, Tamara C. Bidone and Paul A. Sigala, 28 October 2025, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2513845122
The work was supported by the National Institutes of Health (grant numbers R35GM133764, R21AI185746, R35GM14749, and T32AI055434), the Utah Center for Iron & Heme Disorders (grant number U54DK110858), the Price College of Engineering at the University of Utah, and the 3i Initiative at University of Utah Health.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
This is the pure science I subscribed for! (No political spin, narratives or agendas)
The presence of iron crystals in the parasite suggest a way to treat malaria. Place the patient in an alternating magnetic field, causing the iron crystals to oscillate and tear the cells of the parasite apart. Alternatively, it can cause heating of the iron particles and kill the cells that way.
The secret seems to have been discovered. Well done to the team of researchers.