
Researchers have used visible light and gold-copper electrodes to improve CO2 conversion efficiency and selectivity, paving the way for sustainable energy advancements.
By combining visible light with electrochemistry, researchers have significantly improved the conversion of carbon dioxide into valuable products while making a surprising discovery. The team observed that visible light greatly enhanced an important chemical property known as selectivity. This breakthrough not only advances CO2 conversion techniques but also holds promise for a wide range of chemical reactions in catalysis research and chemical manufacturing.
One way that chemists recycle CO2 into valuable products is through a process called electrochemical reduction, where a stream of CO2 gas moves through an electrolysis cell that breaks the CO2 and water down into carbon monoxide and hydrogen, which then can be used to make new desired hydrocarbon products, said University of Illinois Urbana-Champaign chemistry professor Prashant Jain. “However, the reaction is sluggish, and the process requires large electrodes containing a lot of expensive catalyst material such as gold or copper, so our lab has been pursuing ways to speed up the process so that less catalyst material is required, making it a more viable option for the alternative fuels industry.”
The new study, led by Jain and former graduate student Francis Alcorn and published in the Proceedings of the National Academy of Sciences, details a method that combines the action of visible light with electrodes coated in nanoparticles of gold-copper alloy to induce CO2 reduction at a much higher rate and allow for more controlled selectivity than seen with current methods.
Harnessing Visible Light for Electrochemical Reactions
“These new electrodes act like tiny antennae that seek out photons in the visible light range and couple them with the chemical reaction pathway,” Jain said.
In the lab, the electrodes are immersed in a solution of CO2, water, and an electrolyte to enhance conductivity. The team then applies a voltage across the electrode while a visible light laser illuminates its surface. The resulting reaction rapidly produces carbon monoxide — from splitting the CO2 — and hydrogen, which comes from splitting water molecules.
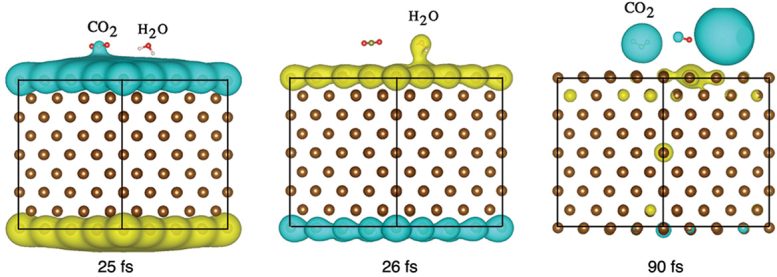
“We were very excited to see the boost in productivity when visible light was used. However, we were not expecting to find that using visible light would have a major impact on chemical selectivity, which is the important advance here,” Jain said.
In catalysis, chemical selectivity is the ability of a chemical reaction to favor or target one type of pathway or molecule over another. In this study, the researchers found that the water-splitting reaction that forms hydrogen gas was selectively enhanced by using light. This led the team to experiment further and model their results with the help of Northwestern University chemistry professor George Schatz and postdoctoral researcher Sajal Kumar Giri.
New Opportunities for Synthetic Gas Production
“The results revealed that visible light offers a unique opportunity to adjust the ratio of carbon monoxide to hydrogen gas produced, a crucial factor for the industrial production of synthetic gas,” Jain said. “This finding paves the way for a more sustainable and efficient energy future.”
Using light to help boost chemical reactions is not without its controversy, though, Jain said. Because adding light to a chemical reaction will also add heat, it was essential for the team to run careful measurements and control experiments to determine if it was simply the heating effect of light that led to faster reaction rates and selectivity.
“We ran experiments with and without the laser at the exact same temperature produced by light excitation and ruled out heating as being responsible,” Jain said. “Rather, electric fields and directed charge flow induced by light excitation were responsible for the enhanced productivity and increased selectivity of water splitting, which is captured in the simulations by our collaborators,” Jain said.
The team still has some challenges to face as they forge ahead. For instance, the repeated use of the nanoparticle-based electrode will inevitably lead to degradation over time, especially under a scaled-up scenario required for industrial application. Additionally, the overall energy efficiency of the process and light management will need further research and improvement.
“What we found with this study presents completely new ways of thinking about electrochemistry and catalysis,” Jain said. “By using light, we enhance the activity of this catalyst, but surprisingly, we also change the selectivity. This will open up new chemical pathways that make different products. And why stop at CO2 reduction or water splitting? This could be applied to many other catalytic reactions important to the chemical industry.”
Reference: “Switching of electrochemical selectivity due to plasmonic field-induced dissociation” by Francis M. Alcorn, Sajal Kumar Giri, Maya Chattoraj, Rachel Nixon, George C. Schatz and Prashant K. Jain, 2 October 2024, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2404433121
Illinois Researchers Maya Chattoraj and Rachel Nixon also contributed to this study. The National Science Foundation, the U.S. Department of Energy, the Robert C. and Carolyn J. Springborn Endowment, and the Future Interdisciplinary Research Explorations Grant supported this research.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Til 2035, when the next cycle of carbon dioxide reduction is reached, due these studies there is hope now to reach the targets.