
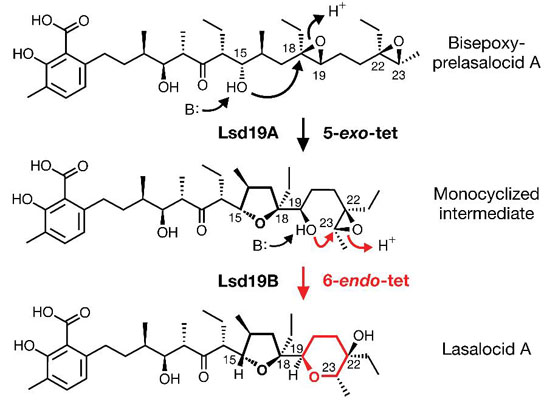
A new study could enable scientists to synthesize many important chemicals currently found only in nature. The research shows how scientists used powerful X-rays to help decipher how five carbon atoms and one oxygen atom in the structure of lasalocid can link into a six-membered ring through an energetically unfavorable chemical reaction.
Researchers working at the U.S. Department of Energy’s (DOE) SLAC National Accelerator Laboratory have used powerful X-rays to help decipher how certain natural antibiotics defy a longstanding set of chemical rules – a mechanism that has baffled organic chemists for decades.
Their result, reported in the journal Nature, details how five carbon atoms and one oxygen atom in the structure of lasalocid, a natural antibiotic produced by bacteria in soil, can link into a six-membered ring through an energetically unfavorable chemical reaction. Unlocking this chemical pathway could enable scientists to synthesize many important chemicals currently found only in nature.
“Our study has a broad implication because the six-membered ring is a common structural feature found in hundreds of drug molecules produced by nature,” said the study’s principal investigator, Chu-Young Kim of the National University of Singapore. “We have actually analyzed the genes of six other organisms that produce similar drugs and we are now confident that the chemical mechanism we have uncovered applies to these other organisms as well.”
According to “Baldwin’s Rules for Ring Closure,” which govern the way these rings form, this compound should contain a five-membered ring instead of the observed six-membered ring.
The solution to the molecular mystery depended in large part on a deeper understanding of the unique protein Lsd19, the catalyst that enables the formation of lasalocid’s rings. To determine the protein’s atomic structure, the researchers hit frozen crystals of Lsd19 with X-rays from SLAC’s Stanford Synchrotron Radiation Lightsource and observed how the crystals diffracted the X-rays passing through. “You need atomic-level detail of the crystal’s structure to understand what’s really happening,” said co-author Irimpan Mathews, a staff scientist at SLAC.
“The bugs have taught us a valuable chemistry lesson,” Kim said. “With a new understanding of how nature synthesizes the six-membered rings, chemists may be able to develop novel methods that will enable us to produce these drugs with ease in the chemical laboratory. Alternatively, protein engineers may be able to use our results to develop a biofactory where these drugs are mass produced using a fermentation method. Either method will make more effective and more affordable drugs available to the public.”
Kim’s group has moved on to their next challenge: investigating how nature synthesizes the anti-cancer drug echinomycin. In the meantime, “The knowledge we have generated will help researchers in academia and industry to develop new methods for biological production of important polyether drugs,” he said. “We are not talking about the distant future.”
Reference: “Enzymatic catalysis of anti-Baldwin ring closure in polyether biosynthesis” by Kinya Hotta, Xi Chen, Robert S. Paton, Atsushi Minami, Hao Li, Kunchithapadam Swaminathan, Irimpan I. Mathews, Kenji Watanabe, Hideaki Oikawa, Kendall N. Houk and Chu-Young Kim, 4 March 2012, Nature.
DOI: 10.1038/nature10865
Additional authors included Kinya Hotta, Xi Chen, Hao Li, and Kunchithapadam Swaminathan of the National University of Singapore, Robert S. Paton of Oxford University, Atsushi Minami and Hideaki Oikawa of Hokkaido University, Kenji Watanabe of the University of Shizuoka and Kendall N. Houk of the University of California Los Angeles. The research was supported by the Royal Commission for the Exhibition of 1851 and Fulbright-AstraZeneca Research Fellowship, the Japan Society for the Promotion of Science, the National Institutes of Health, the Ministry of Education, Culture, Sports, Science & Technology in Japan, and the National University of Singapore Life Sciences Institute Young Investigator Award. The Stanford Synchrotron Radiation Lightsource is supported by DOE’s Office of Science and its Structural Molecular Biology program is also supported by the National Institutes of Health.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.