Viewing Proteins at the Nanoscale Offers Insights for Treating Neurodegenerative Diseases
Many diseases affecting the brain and nervous system are linked to the formation of protein aggregates, or solid condensates, in cells from their liquid form condensate, however, little is known about this process.
This liquid-to-solid transition can trigger the formation of what are called amyloid fibrils. These can further form plaques in neurons causing neurodegenerative diseases such as Alzheimer’s.
Biomedical engineers at the University of Sydney, in collaboration with scientists at the University of Cambridge and Harvard University, have now developed sophisticated optical techniques to monitor at close range the process by which these protein aggregates form.
By testing a protein associated with Amyotrophic Lateral Sclerosis – ALS disease, which affected astrophysicist Professor Stephen Hawking – the Sydney engineers closely monitored the transition of this protein from its liquid to solid phase.
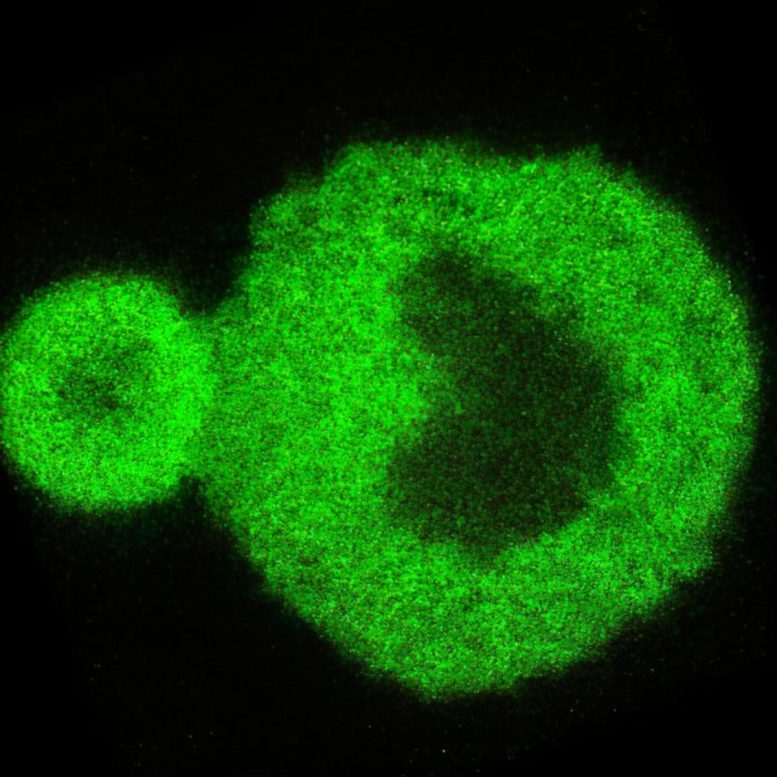
3D confocal microscopy scan of a FUS protein condensate incubated for 24 hours showing the characteristic core-shell structure unveiled by this research. Credit: The University of Sydney
“This is a huge step forward to understanding how neurogenerative diseases develop from a fundamental perspective,” said Dr Yi Shen, lead author of the research published in the Proceedings of the National Academy of Sciences (PNAS) in the United States.
“We can now directly observe the transition of these critical proteins from liquid to solid at the nanoscale – a millionth of a meter in scale,” said Dr. Daniele Vigolo, a senior lecturer in the School of Biomedical Engineering and a member of the University of Sydney Nano Institute.
The Dual Role of Protein Condensates
Proteins regularly form condensates during liquid-to-liquid phase separation in a wide range of critical and healthy biological functions, such as the formation of human embryos. This process assists biochemical reactions where protein concentrations are critical and also promotes healthy protein–protein interactions.

How Protein Aggregation Contributes to Disease
“However, this process also increases the risk of dysfunctional aggregation, where unhealthy aggregates of solid proteins form in human cells,” said Dr Shen, who is an ARC DECRA Fellow in the School of Chemical and Biomolecular Engineering and also a member of Sydney Nano.
“This can lead to aberrant structures associated with neurodegenerative diseases because the proteins no longer exhibit rapid reversibility back to liquid form. It is therefore crucial to monitor condensate dynamics, as they directly affect pathological states,” she said.
The world-first nanoscale optical observation of this process has allowed the team to determine that the transition from liquid to solid protein starts at the interface of the protein condensates. This window onto the phase transition also revealed that the internal structures of these protein agglomerates are heterogenous, where previously they were thought to be homogeneous.
Dr Vigolo said: “Our findings promise to greatly improve our understanding of neurogenerative diseases from a fundamental perspective.
“This means a promising new area of research to better understand how Alzheimer’s disease and ALS develops in the brain, affecting millions of people worldwide.”
Reference: “The liquid-to-solid transition of FUS is promoted by the condensate surface” by Yi Shen, Anqi Chen, Wenyun Wang, Yinan Shen, Francesco Simone Ruggeri, Stefano Aime, Zizhao Wang, Seema Qamar, Jorge R. Espinosa, Adiran Garaizar, Peter St George-Hyslop, Rosana Collepardo-Guevara, David A. Weitz, Daniele Vigolo and Tuomas P. J. Knowles, 7 August 2023, Proceedings of the National Academy of Sciences.
DOI: 10.1073/pnas.2301366120
The study was funded by the Frances and Augustus Newman Foundation, the Wellcome Trust, the European Research Council, the US Alzheimer Association, ALS Canada-Brain Canada, the Canadian Institutes of Health Research, and the National Institute on Aging.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.