
Neocapritermes taracua termites carry a life-ending enzyme that, when mixed with another compound during attacks, produces a lethal liquid, sacrificing the termite to protect its colony.
Older worker termites of the species Neocapritermes taracua protect their colonies with an unparalleled defense mechanism. When the colony is attacked, they sacrifice themselves by setting off an explosive chemical reaction, the result of which is a toxic liquid that immobilizes and poisons their adversary.
Now, researchers from the Institute of Organic Chemistry and Biochemistry of the Czech Academy of Sciences, in cooperation with colleagues from the Faculty of Tropical AgriScience of the Czech University of Life Sciences in Prague, have unraveled the mysteries of these kamikaze termites.
In a study published in the scientific journal Structure, Dr. Jana Škerlová and her colleagues from the scientific group of Assoc. Prof. Pavlína Maloy Řezáčová provide a detailed description of the mechanism by which the mysterious enzyme that termites carry on their backs works.
Researchers from the Institute of Organic Chemistry and Biochemistry of the Czech Academy of Sciences, in cooperation with colleagues from the Faculty of Tropical AgriScience of the Czech University of Life Sciences in Prague, are unraveling the mysteries of the life of termites.
Unique Defense Mechanisms of Neocapritermes Taracua
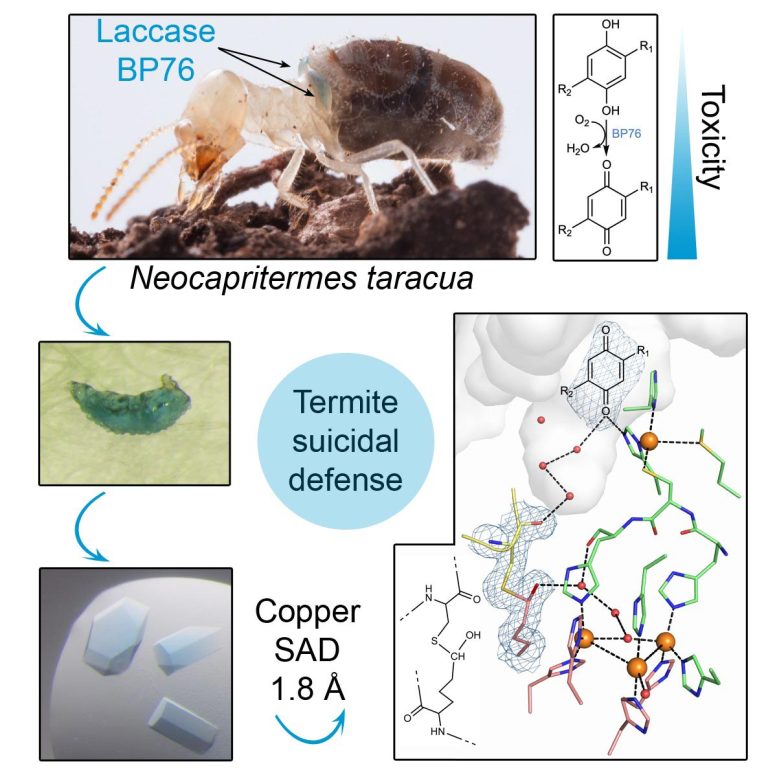
The termite species Neocapritermes taracua has evolved a peculiar defense mechanism that is unparalleled in the insect world. Worker termites play a key role in it. Over their lifetime, they gradually amass a particular enzyme, blue laccase BP76, in special pockets on their backs. When their colony finds itself in danger, older individuals tear this ‘rucksack’ apart. The enzyme is then almost immediately mixed with another substance stored in the termite body, which up to this point is relatively harmless, creating a sticky liquid containing highly poisonous benzoquinones. Although this kills the kamikaze termite itself, it also immobilizes or kills the attacker.

Scientific Breakthrough in Enzyme Stability
How this potentially explosive enzyme stays active in a solid state on the backs of insects was a true scientific riddle. Scientists from the Structural Biology research group at IOCB Prague have solved the puzzle with the help of X-ray crystallography. Jana Škerlová was intrigued by the fact that the blue laccase borne by termites contains an unusually strong bond between two amino acids—which are the building blocks of proteins—near the active site of the enzyme, to which the target molecule binds and where it reacts.
She explains: “Unravelling the three-dimensional structure of laccase BP76 revealed that this enzyme uses a variety of stabilization strategies, which make it not only highly durable, but also fully functional even in the harsh conditions of tropical rainforests.”
Due to its unique structure, laccase BP76 not only remains intact but also active even though it rests on the back of a termite throughout its entire life. This is crucial for the enzyme’s role in the defense mechanism because, in the event of an attack on the colony, the reaction must be immediate.
Lifetime Burden of Termites
Termites of the species Neocapritermes taracua can live a whole lifetime with this suicidal load. Young individuals, who are still capable of doing a lot of work for their colony, carry only small amounts of the enzyme in their back pockets. The blue ‘rucksack’, in which the explosive material accumulates, grows larger over time as the insect loses strength. Its last service to the termite mound is that it is prepared to sacrifice itself for the good of the colony.

Role of Structural Biology in Understanding Termite Defense
The fact that Neocapritermes taracua termites have solid packets of an active enzyme tucked into pockets of their raincoats, which they do not hesitate to use as a weapon in an emergency, was first observed by researchers in French Guiana some years ago. That research, published in the journal Science, also bears the IOCB Prague seal. One of the researchers who collaborated on the seminal study was Professor Jan Šobotník, who is also a co-author of the present paper and currently works at the Faculty of Tropical AgriScience of the Czech University of Life Sciences.
“Our discovery is an excellent illustration of the irreplaceable role of structural biology. Just as knowledge about individual components of an instrument sheds light on how it works, knowing the three-dimensional structure (i.e. the positions of individual atoms) of a molecule helps us understand a biological process. In this case, it is a unique defense mechanism of termites,” emphasizes Pavlína Řezáčová, head of the laboratory from which the research originates.
Reference: “Crystal structure of blue laccase BP76, a unique termite suicidal defense weapon” by Jana Škerlová, Jiří Brynda, Jan Šobotník, Marek Zákopčaník, Petr Novák, Thomas Bourguignon, David Sillam-Dussès and Pavlína Řezáčová, 15 August 2024, Structure.
DOI: 10.1016/j.str.2024.07.015
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
Looks like Hezbollah as been eating Lebanese bush-termites and are blowing themselves up.