
Scientists have uncovered an unexpected way that certain bacteria manipulate plants from within.
Plant-infecting bacteria have a surprisingly direct way of taking over crops. Instead of slowly breaking down defenses, many of them inject proteins straight into plant cells, effectively hijacking the system from the inside.
For decades, scientists have tried to understand one particularly important group of these proteins, known as AvrE/DspE. These molecules are used by pathogens that attack a wide range of crops, including rice, tomatoes, apples, and pears. They are responsible for diseases such as bacterial speck, brown spot, and the devastating fire blight that can wipe out entire orchards.
Unraveling a Long-Standing Mystery
Now, in findings published in Nature, researchers have uncovered how these mysterious proteins actually function, revealing a mechanism that could open the door to new ways of protecting crops.
Despite being identified in the early 1990s, these proteins have remained a mystery. Researchers knew they were essential for infection because disabling them renders harmful bacteria harmless. They also knew the proteins could suppress plant immune responses and trigger early symptoms like water-soaked lesions on leaves. What they could not explain was how these proteins physically worked inside plant cells.

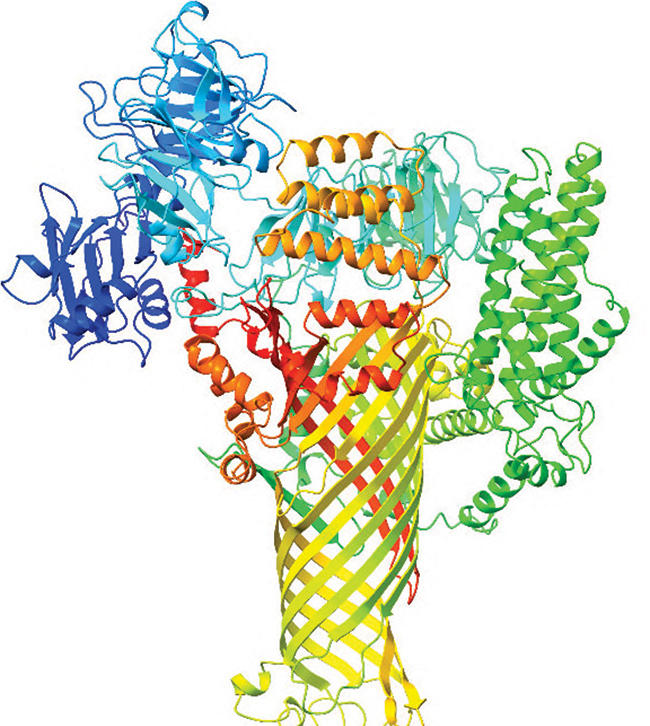
These proteins are unusually large, containing about 2,000 amino acids compared to roughly 300 in a typical bacterial protein. That size made them hard to study with traditional methods, and comparisons to known proteins offered few clues.
“They’re weird proteins,” said biologist Sheng-Yang He.
A Structural Breakthrough
A breakthrough came with AlphaFold2, an artificial intelligence tool that predicts protein structures. When researchers used it to model AvrE/DspE proteins, they found a surprising shape. The proteins appeared to form hollow, tube-like structures, similar to tiny straws. This matched cryo-electron microscopy images of a fire blight protein, which also showed a hollow form.
“When we first saw the model, it was nothing like what we had thought,” said study co-author Pei Zhou of Duke University.
The structure suggested a new role. Instead of only weakening plant defenses, these proteins may act as channels that pull water into plant tissue.
Explaining a common symptom of infection
Leaves often become waterlogged, creating a moist environment where bacteria can grow. Normally, the apoplast, the space between plant cells, stays relatively dry to support photosynthesis. During infection, that balance shifts.
Further analysis showed the proteins have a water-repelling exterior but a water-attracting inner channel, supporting the idea that they guide water into plant tissue.
To test this, researchers engineered frog eggs to produce the proteins. When placed in a dilute saline solution, the eggs rapidly absorbed water, swelled, and burst, consistent with the channel hypothesis.
The team then tried to block the process using PAMAM dendrimers, tiny nanoparticles with precisely controlled sizes.
“We were tinkering with the hypothesis that if we found the right diameter chemical, maybe we could block the pore,” He said.
They identified a particle that could plug the channel formed by the fire blight pathogen Erwinia amylovora. Treated frog eggs no longer swelled. In plants, the results were just as striking. Infected Arabidopsis showed a 100-fold drop in bacterial levels, and pear fruits exposed to fire blight bacteria stayed healthy.
“It was a long shot, but it worked,” He said. “We’re excited about this.”
Toward New Crop Protection Strategies
Because many plant pathogens rely on these proteins, the findings point to a potential broad-spectrum way to protect crops. Instead of killing bacteria directly, this approach blocks a key mechanism they use to survive inside plants.
That could have major implications. Crops provide about 80% of the food humans consume, yet more than 10% of global production is lost each year to pests and disease, costing an estimated $220 billion.
The researchers have filed a provisional patent and are now working to better understand how the nanoparticles interact with the protein channels, with the goal of developing more effective crop protection strategies.
“If we can image those structures we can have a better understanding and come up with better designs for crop protection,” Zhou said.
A related follow-up study published in Trends in Microbiology further strengthens these findings by showing that the AvrE/DspE protein family is not only widespread but also remarkably conserved across many of the world’s most damaging plant pathogens.
These effectors are shared by bacteria that infect a wide range of crops, highlighting their central role in disease. Researchers confirmed that these proteins do more than suppress plant defenses. They actively reshape plant physiology by inducing water-soaking, disrupting immune signaling, and even triggering cell death in some cases.
References: “Bacterial pathogens deliver water- and solute-permeable channels to plant cells” by Kinya Nomura, Felipe Andreazza, Jie Cheng, Ke Dong, Pei Zhou and Sheng Yang He, 13 September 2023, Nature.
DOI: 10.1038/s41586-023-06531-5
“The conserved AvrE family of bacterial effectors: functions and targets during pathogenesis” by Laura Herold, Sera Choi, Sheng Yang He and Cyril Zipfel, 14 September 2024, Trends in Microbiology.
DOI: 10.1016/j.tim.2024.08.007
The study was funded by the National Institute of Allergy and Infectious Diseases and the National Institute of General Medical Sciences, both at the National Institutes of Health, Duke Science and Technology, and the Howard Hughes Medical Institute.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.