Scientists demonstrate new method of producing a specific class of organic compounds, which promises to accelerate drug discovery research for several diseases.
Several drugs, including those for depression, schizophrenia, and malaria, would not be if not for a type of organic chemical compound called alicyclic compounds. These compounds are 3D structures formed when three or more carbon atoms join in a ring via covalent bonds, but the ring is not aromatic.
Aromatic compounds (or arenes) are another class of organic compounds which are 2D structures with reactive properties distinct from those of alicyclic compounds. A well-known example is benzene, the six-carbon ring comprising alternating single- and double-bonds between the carbon atoms.
By dearomatizing arenes, one can get alicyclic compounds. In fact, this dearomatization is one of the most powerful ways of obtaining alicyclic compounds. But some of the most abundantly available arenes, such as benzene and naphthalene, are very stable, and breaking them up to construct alicyclic compounds has been challenging. With existing methods, often large amounts of reactants yield very little product.
“The highly efficient conversion of readily and commercially available arenes to high value-added alicyclic compounds could accelerate drug discovery research by leaps,” said Assistant Professor Kei Muto and Professor Junichiro Yamaguchi of Waseda University, Japan, who led the discovery of a novel efficient method. Their study is published in the Royal Society of Chemistry’s Chemical Science.
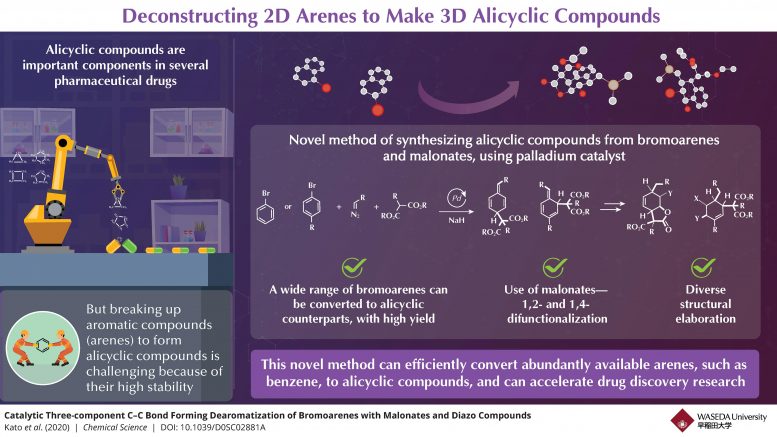
In the novel method, bromoarenes are reacted with two other classes of organic compounds, diazo compounds, and malonates, in the presence of a palladium catalyst (a compound that enables a chemical reaction), under optimal conditions of concentration, temperature, and time (experimentally ascertained in the study). Subsequently, good amounts of the corresponding alicyclic compounds are produced.
“What is really special about this method is that a range of bromoarenes, including benzenoids, azines, and heteroles, can be converted to their alicyclic counterparts,” Muto says. He speaks also of the key portions of an alicyclic molecule that give it complexity and utility–the functional groups attached to the cyclic carbons. He says, “The obtained compounds have functional groups at two points in the cyclic chain, and these can be easily diversified through further reactions to yield a variety of highly functionalized 3D molecules.”
The use of malonates as reactant is what allows this multi-functionalization, setting this novel method apart from existing methods, which are often highly specific in terms of the products possible. Because malonates are known to predominantly react with palladium-benzyl complexes, the use of a palladium-based catalyst became key to the success of this method. The palladium catalyst led to the formation of a benzyl-palladium intermediate that could then react with malonates, producing the final multi-functionalized alicyclic products.
Thus, designing an appropriate catalysis process was essential to developing the aromatic-to-alicyclic transformation technique. “Next, we would like to design new catalysts to make this reaction more general; that is, compatible with a broader range of arenes,” says Yamaguchi.
With their future plans in place, Muto and Yamaguchi are confident of the good that their team’s work can do in the world: “We believe this organic reaction will help drug discovery research finally ‘escape from the flatland’ of the simpler and 2D aromatic compounds, so to speak, thereby advancing medicinal chemistry significantly.”
Reference: “Catalytic three-component C–C bond forming dearomatization of bromoarenes with malonates and diazo compounds” by Hiroki Kato, Itsuki Musha, Masaaki Komatsuda, Kei Muto and Junichiro Yamaguchi, 29 July 2020, Chemical Science.
DOI: 10.1039/D0SC02881A
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.