A gut bacterium, Coprococcus comes, may reduce the effectiveness of ACE inhibitors, leading to treatment-resistant hypertension. This finding could help develop personalized treatments based on gut microbial composition.
Almost half of the U.S. adult population has high blood pressure — or hypertension — and about 20% of these patients have treatment-resistant hypertension. The reason why some people are resistant to treatment has been a mystery, but new study results show that a certain gut bacterium may be an important factor.
“Today, doctors treat resistant hypertension by adding or substituting medications, which can contribute to overdoses, more side effects, and noncompliance,” said Tao Yang, PhD, assistant professor at the University of Toledo. “A better understanding of the relationship between gut microbes and drug efficacy could lead to new treatment approaches for people who don’t respond to blood pressure medication. This could include new drugs or modulating gut microbiota with probiotics, antibiotics, and other methods.”
By studying the gut microbes of rats, Yang and colleagues discovered that a bacteria known as Coprococcus comes contributes to resistance to ACE inhibitors, one of the primary drug classes used to treat high blood pressure.
Coprococcus Comes and ACE Inhibitor Resistance
Yang will present the new research at the American Society for Pharmacology and Experimental Therapeutics annual meeting during the Experimental Biology (EB) 2022 meeting, to be held on April 2-5, 2022, in Philadelphia.
“Our ultimate goal is to find a link between gut microbial composition and enzymatic activity and drug response effectiveness because this will provide a foundation for applying precision medicine to treat resistant hypertension,” Yang said.
Research has shown that the microorganisms in our gut — collectively known as the gut microbiota — contain a variety of enzymes that can affect drug metabolism.
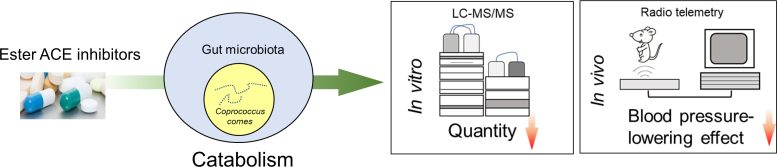
To find out if gut microbiota might play a role in resistance to blood pressure medicine, Yang and colleagues administered a single dose of the ACE inhibitor quinapril to rats with high blood pressure. They found that quinapril was more effective at lowering blood pressure in hypertensive rats with a lower gut microbiota load. When they analyzed the composition of the gut microbiota, C. comes emerged as an important player.
Through additional experiments, the researchers found that C. comes can actually break down quinapril. They also observed that giving C. comes and quinapril to hypertensive rats reduced blood pressure less than administering quinapril alone.
Future Directions in Precision Medicine
“We are still in the early stages of determining the interactions between gut bacteria and antihypertensive medications,” said Yang. “However, our current findings suggest that the same drug may not be appropriate for everyone because each person has a unique gut microbial composition with a unique profile of enzymatic activities.”
The researchers are now performing similar experiments using other types of gut bacteria and additional blood pressure medications to further explore how the gut microbiota modulates the effectiveness of antihypertensive drugs.
Tao Yang will present this research on from 10 a.m.–12 p.m., Tuesday, April 5, in Exhibit/Poster Hall A-B, Pennsylvania Convention Center (Poster Board Number B166) (abstract). Contact the media team for more information or to obtain a free press pass to attend the meeting.
Meeting: Experimental Biology 2022
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.