A fast, green and one-step method for producing porous carbon spheres, which are a vital component for carbon capture technology and for new ways of storing renewable energy, has been developed by Swansea University researchers.
The method produces spheres that have good capacity for carbon capture, and it works effectively at a large scale.
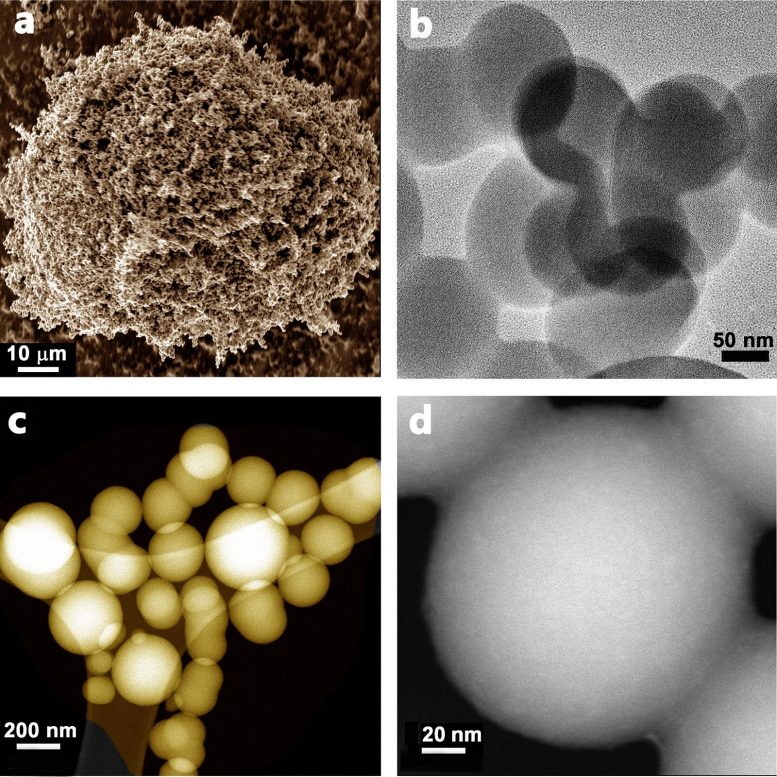
Carbon spheres range in size from nanometers to micrometers. Over the past decade they have begun to play an important role in areas such as energy storage and conversion, catalysis, gas adsorption and storage, drug and enzyme delivery, and water treatment.
They are also at the heart of carbon capture technology, which locks up carbon rather than emitting it into the atmosphere, thereby helping to tackle climate change.
The problem is that existing methods of making carbon spheres have drawbacks. They can be expensive or impractical, or they produce spheres that perform poorly in capturing carbon. Some use biomass, making them more environmentally friendly, but they require a chemical to activate them.
This is where the work of the Swansea team, based in the University’s Energy Safety Research Institute, represents a major advance. It points the way towards a better, cleaner, and greener way of producing carbon spheres.
The team adapted an existing method known as CVD — chemical vapor deposition. This involves using heat to apply a coating to a material. Using pyromellitic acid as both carbon and oxygen source, they applied the CVD method at different temperatures, from 600-900 °C. They then studied how efficiently the spheres were capturing CO2 at different pressures and temperatures
They found that:
- 800 °C (1,472 °F) was the optimum temperature for forming carbon spheres
- The ultramicropores in the spheres that were produced gave them a high carbon capture capacity at both atmospheric and lower pressures
- Specific surface area and total pore volume were influenced by the deposition temperature, leading to an appreciable change in overall carbon dioxide capture capacity
- At atmospheric pressure the highest CO2 adsorption capacities, measured in millimolars per gram, for the best carbon spheres, were around 4.0 at 0 °C (32 °F) and 2.9 at 25 °C (77 °F).
This new approach brings several advantages over existing methods of producing carbon spheres. It is alkali-free and it doesn’t need a catalyst to trigger the shaping of the spheres. It uses a cheap and safe feedstock that is readily available in the market. There is no need for solvents to purify the material. It is also a rapid and safe procedure.
Dr. Saeid Khodabakhshi of the Energy Safety Research Institute at Swansea University, who led the research, said:
“Carbon spheres are fast becoming vital products for a green and sustainable future. Our research shows a green and sustainable way of making them.
We demonstrated a safe, clean, and rapid way of producing the spheres. Crucially, the micropores in our spheres means they perform very well in capturing carbon. Unlike other CVD methods, our procedure can produce spheres at large scale without relying on hazardous gas and liquid feedstocks.
Carbon spheres are also being examined for potential use in batteries and supercapacitors. So in time, they could become essential to renewable energy storage, just as they already are for carbon capture.”
The research was published in the journal Carbon.
Reference: “Facile and environmentally friendly synthesis of ultramicroporous carbon spheres: A significant improvement in CVD method” by Saeed Khodabakhshi, Sajad Kiani, Yubiao Niu, Alvin Orbaek White, Wafa Suwaileh, Richard E. Palmer, Andrew R. Barron and Enrico Andreoli, 1 September 2020, Carbon.
DOI: 10.1016/j.carbon.2020.08.056
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.