By using the Linac Coherent Light Source at Stanford to analyzed tiny protein crystals, scientists demonstrated the potential of free-electron lasers for obtaining the structures of macromolecules from tiny crystals and their importance as a new tool for structural biology on large macromolecular assemblies and membrane proteins.
In the centennial year of Max von Laue’s discovery that X-ray diffraction can be used to unravel the atomic architecture of molecules, a new approach to the determination of high-resolution structures has been demonstrated. An international team of researchers has analyzed tiny protein crystals using short pulses of X-ray light from the world’s first hard X-ray free-electron laser, the US Department of Energy’s 300 million dollar Linac Coherent Light Source at Stanford.
The study demonstrates the immense potential of free-electron lasers for obtaining the structures of macromolecules from tiny crystals when illuminated with the blazing intensity of the ultrashort free-electron laser X-ray pulses, even though the crystals are destroyed in the process. In the current study, their structural analysis reveals details with a spatial resolution of 0.2 millionth of a millimeter. The team, including researchers from the Max Planck Institute for Medical Research in Heidelberg and the Max Planck Advanced Study Group in Hamburg, showed that their data compared well with those collected from large, well-characterized crystals using conventional X-ray sources, providing a benchmark for the new free-electron laser approach. Their proof-of-principle experiment shows that the free-electron laser is an important new tool for structural biology on large macromolecular assemblies and membrane proteins, many of which are known to be important targets for pharmaceutical development.
X-ray free-electron lasers are extremely powerful new X-ray sources that provide highly intense ultrashort flashes of light. The intensity of such an X-ray pulse is more than a billion times higher than that provided by the most brilliant state-of-the-art X-ray sources, with a thousand-fold shorter pulse length, on the order of a few millionths of a billionth of a second, or femtoseconds. These properties provide scientists with novel tools to explore the nano-world, including the structure of biological materials.
Most of our knowledge of the three-dimensional spatial architecture of molecules has been obtained by X-ray crystallography, which relies on the amplification of the scattering signal of the molecules by their arrangement into relatively large crystals, often on the order of some tenths of a millimeter. Obtaining large crystals can be extremely difficult in the case of bio-molecules due to their inherent instability and flexibility, as well as their typical low abundance.
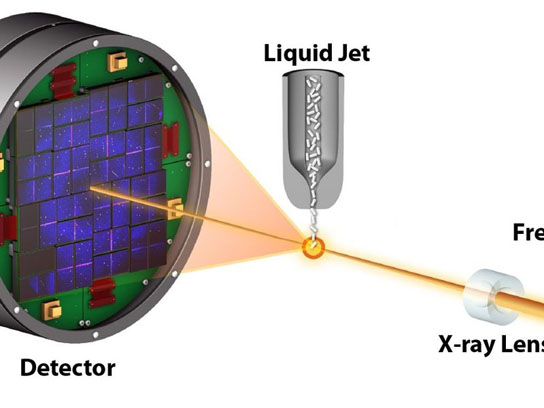
Free-electron lasers can obtain structural information from tiny crystals that refuse to reveal their secrets by conventional structural methods due to the damage induced by the radiation used for the structure analysis. Although the tiny crystals are completely destroyed by the incredibly intensity of the free-electron laser, the ultra-short pulses can pass through the sample before the onset of detectable damage and thus provide the necessary scattering signal of the still-intact molecules.
In this diffraction-before-destruction approach, crystals are replenished for serial data collection by injecting them into the free-electron laser beam using a liquid jet, developed by scientists from Arizona State University, exposing one crystal after the other instead of rotating a single large crystal as in conventional crystallography. This concept of serial femtosecond crystallography has been demonstrated before by the same team of researchers at the Linac Coherent Light Source at Stanford, using the CAMP instrument, developed by the Max Planck Advanced Study Group. The relatively long wavelength X-rays then available limited the attainable level of structural detail.
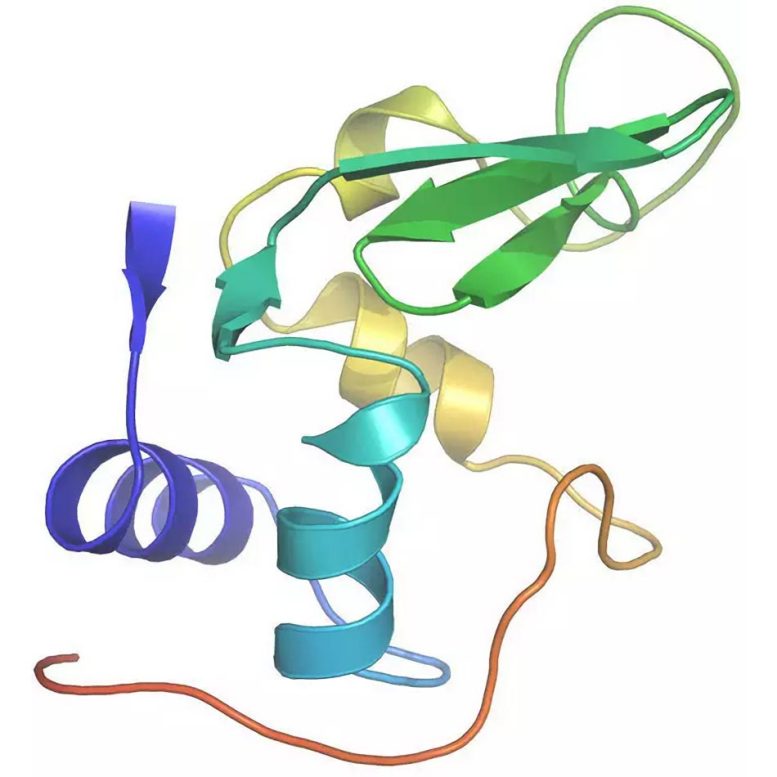
Very recently, a new instrument at the Linac Coherent Light Source, the Coherent X-ray Imaging endstation, has allowed the use of short wavelength X-rays and thus made it possible to infer atomic detail in the molecular architecture. To benchmark the method, a well-characterized model system was investigated, the small protein lysozyme, the first enzyme ever to have its structure revealed.
The researchers collated ten thousand snapshot exposures from crystals that measured only a thousandth of a millimeter, and showed that the data compared well with those collected using conventional approaches and hundred-fold larger lysozyme crystals. Importantly, no significant signs of radiation damage were detected. “This proof-of-principle experiment shows that the X-ray free-electron laser indeed lives up to its promise as an important new tool for structural biology on large macromolecular assemblies and membrane proteins. It really opens up a completely new terrain in structural biology,” Ilme Schlichting, leading the Max-Planck team, says. Since small crystals are typically easier to produce than large ones, this is of immediate relevance for all studies of molecules that are difficult to crystallize — as are some 60 percent of all proteins, many of which are prime targets for medical therapies.
Reference: “High-Resolution Protein Structure Determination by Serial Femtosecond Crystallography” by Sébastien Boutet, Lukas Lomb, Garth J. Williams, Thomas R. M. Barends, Andrew Aquila, R. Bruce Doak, Uwe Weierstall, Daniel P. DePonte, Jan Steinbrener, Robert L. Shoeman, Marc Messerschmidt, Anton Barty, Thomas A. White, Stephan Kassemeyer, Richard A. Kirian, M. Marvin Seibert, Paul A. Montanez, Chris Kenney, Ryan Herbst, Philip Hart, Jack Pines, Gunther Haller, Sol M. Gruner, Hugh T. Philipp, Mark W. Tate, Marianne Hromalik, Lucas J. Koerner, Niels van Bakel, John Morse, Wilfred Ghonsalves, David Arnlund, Michael J. Bogan, Carl Caleman, Raimund Fromme, Christina Y. Hampton, Mark S. Hunter, Linda C. Johansson, Gergely Katona, Christopher Kupitz, Mengning Liang, Andrew V. Martin, Karol Nass, Lars Redecke, Francesco Stellato, Nicusor Timneanu, Dingjie Wang, Nadia A. Zatsepin, Donald Schafer, James Defever, Richard Neutze, Petra Fromme, John C. H. Spence, Henry N. Chapman and Ilme Schlichting, 31 May 2012, Science.
DOI: 10.1126/science.1217737
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.