As the antimalarial drugs hydroxychloroquine and chloroquine have drawn attention as potential therapies for COVID-19 and are being widely used off-label, it’s now more important than ever to have a thorough assessment of the safety of these medications. A recent analysis published in the British Journal of Clinical Pharmacology provides new insights.
In the analysis of real-world data from the Food and Drug Administration Adverse Events Reporting System, a global database of post-marketing safety reports, hydroxychloroquine and chloroquine were associated with higher rates of various cardiovascular problems, including life-threatening heart rhythm events, heart failure, and damage to the heart muscle itself (termed cardiomyopathy).
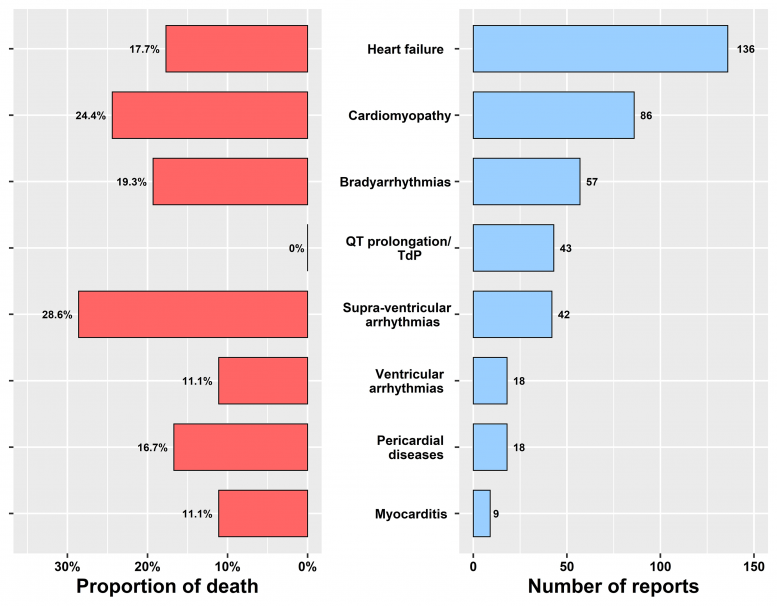
“Moreover, we show how these adverse events carry high risks for severe outcomes including death, even with standard doses of the drugs,” said senior author Elad Maor, MD, PhD, of Sheba Medical Center and Tel-Aviv University, in Israel. “The take-home message of our work is that physicians around the world should be careful when prescribing these drugs for off-label indications, especially for patients with cardiac disorders.”
Reference: “Cardiovascular adverse events associated with hydroxychloroquine and chloroquine: A comprehensive pharmacovigilance analysis of pre‐COVID‐19 reports” by Adam Goldman, David Bomze, Rachel Dankner, Hanoch Hod, Tomer Meirson, Ben Boursi and Elad Maor, 22 September 2020, British Journal of Clinical Pharmacology.
DOI: 10.1111/bcp.14546
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.