
New catalyst developed at Goethe University Frankfurt can break strong carbon-fluorine bonds, with potential applications in pharmaceutical production.
PFAS are remarkable molecules in many respects. Even a thin coating can repel water, oil, and dirt. They are also highly resistant to heat and UV light, making them ideal for products like breathable outdoor clothing, stain-resistant carpets, disposable tableware, irons, and non-stick cookware.
In industrial settings, PFAS are used as lubricants, surfactants, wetting agents, in chrome plating, and in fire-fighting foams. Their applications are widespread, and PFAS are found in countless everyday items.
However, these advantages come with serious drawbacks. Because PFAS are so stable, they remain in the environment long after their use. Although they can be nearly completely destroyed in waste incineration plants, they may still build up during recycling processes—such as in textiles or sewage sludge—and eventually make their way into the environment.

PFAS have been detected in water, soil, plants, and even inside the human body. This is especially concerning because some of the roughly 4,700 known PFAS compounds are suspected to be carcinogenic or linked to other health risks.
How the catalyst works at the molecular level
The key to both the effectiveness and environmental persistence of PFAS lies in their extremely stable molecular structure, particularly the carbon–fluorine (C–F) bonds. A team of chemists led by Professor Matthias Wagner at Goethe University’s Institute of Inorganic and Analytical Chemistry has now developed a catalyst that can break these C–F bonds within seconds at room temperature.
At the core of the catalyst are two boron atoms embedded in a carbon framework, which makes the structure resistant to air and moisture—a rare and highly useful feature for boron-based compounds.
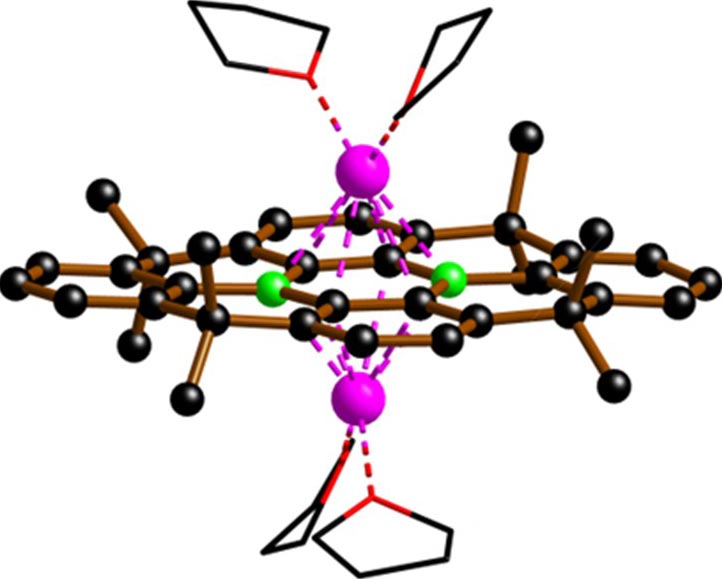
Christoph Buch, a doctoral researcher in Wagner’s group and first author of the study, explains: “To break C–F bonds, we need electrons, which our catalyst transfers with exceptional efficiency. So far, we’ve been using alkali metals like lithium as the electron source, but we’re already working on switching to electrical current instead. That would make the process both much simpler and more efficient.”
Future applications in pharmaceuticals
Beyond PFAS degradation, Wagner sees broader applications for the catalyst: “Many pharmacologically important substances contain fluorine atoms to increase their physiological stability and enhance their effect. Fluorine atoms can also improve drug uptake. With this catalyst, we now have a tool that allows us to precisely control the degree of fluorination in such compounds.”
Reference: “Planarity Is Not Plain: Closed- vs Open-Shell Reactivity of a Structurally Constrained, Doubly Reduced Arylborane toward Fluorobenzenes” by Christoph D. Buch, Alexander Virovets, Eugenia Peresypkina, Burkhard Endeward, Hans-Wolfram Lerner, Felipe Fantuzzi, Shigehiro Yamaguchi and Matthias Wagner, 29 May 2025, Journal of the American Chemical Society.
DOI: 10.1021/jacs.5c05588
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
4 Comments
Could there be a similar catalyst that can break Hydrogen Fluorine Bonds?
Please elaborate.
Really?
And what about your liver?!
Your liver will not break down “forever chemicals”
Down with PFAS