
Scientists created a stem cell-based method to produce high-quality mitochondria at scale, enabling effective treatments for osteoarthritis and other diseases linked to mitochondrial dysfunction.
Scientists have developed a groundbreaking method to mass-produce high-quality human mitochondria, a breakthrough that could revolutionize treatments for degenerative diseases. By optimizing stem cell culture conditions, researchers achieved an 854-fold increase in mitochondrial yield, along with a significant boost in energy output.
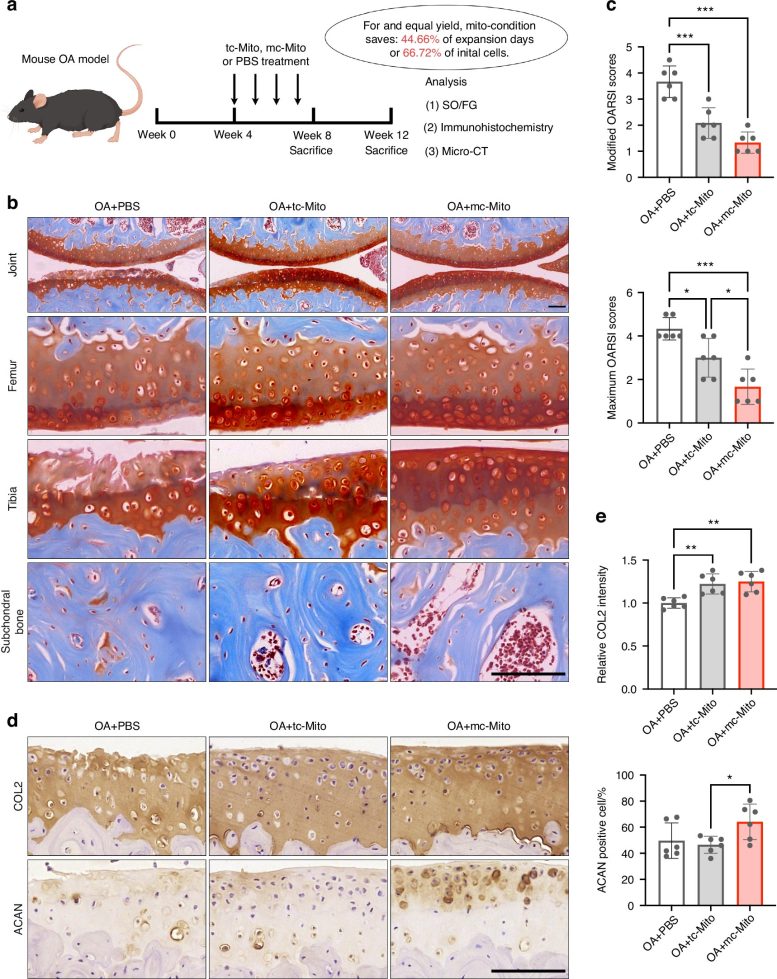
These lab-grown mitochondria demonstrated powerful therapeutic effects, especially in osteoarthritis models, where they accelerated cartilage regeneration. This advancement overcomes a major challenge in mitochondrial transplantation: the limited availability and variable quality of donor mitochondria.
In addition to its clinical potential, the study offers new insights into cellular energy regulation. It shows that cells can be reprogrammed to prioritize mitochondrial production, opening the door to new therapies for a wide range of conditions linked to mitochondrial dysfunction, including joint degeneration and cardiovascular disease.
Mitochondrial dysfunction is a common denominator in numerous diseases, including osteoarthritis, heart failure, and metabolic disorders. While mitochondrial transplantation has emerged as a promising avenue for restoring tissue function, its clinical potential has been severely hampered by the scarcity of viable mitochondria.
Current methods rely on extracting mitochondria from donor tissues, yielding only limited quantities with variable quality, sufficient for a single treatment at best. Moreover, the intricate structure of mitochondria makes synthetic production an enormous challenge.
Given the immense demand—up to one billion mitochondria per patient—existing approaches fall far short of therapeutic needs. Recognizing these limitations, researchers sought a sustainable and scalable solution for producing high-quality mitochondria suitable for clinical applications.
Inside the Mito-Condition Medium
In a new study published in Bone Research, a team from Zhejiang University School of Medicine has pioneered a stem cell-based system that functions as a “mitochondria factory.” By leveraging human mesenchymal stem cells and a specially designed culture medium dubbed “mito-condition,” the team achieved unparalleled increases in both mitochondrial quantity and quality.
These mitochondria exhibited exceptional energy production and facilitated cartilage regeneration in osteoarthritis models. This groundbreaking approach not only overcomes key barriers in mitochondrial transplantation but also deepens our understanding of cellular organelle regulation, opening new frontiers in regenerative medicine.
At the heart of the breakthrough is the innovative “mito-condition” culture medium, which integrates nine essential components, including growth factors and human platelet lysate, to optimize mitochondrial production. Within just 15 days, this method generated 854 times more mitochondria than conventional approaches, all while preserving stem cell viability. The manufactured mitochondria displayed extraordinary functionality, producing 5.7 times more ATP than naturally occurring mitochondria and maintaining stable performance even post-isolation.
Mechanistic studies revealed that the mito-condition medium activates the AMPK pathway, a crucial cellular energy sensor, driving upregulation of mitochondrial biogenesis genes such as TFAM. Remarkably, cells undergoing this process downregulated energy-intensive activities like autophagy and secretion, effectively prioritizing mitochondrial synthesis. Transmission electron microscopy confirmed the unique characteristics of these lab-grown mitochondria, which appeared in a distinct rounded form and were significantly more abundant than their native counterparts.
Therapeutic Impact and Clinical Viability
In osteoarthritis models, transplantation of these enhanced mitochondria resulted in substantial cartilage repair over a 12-week period, surpassing the efficacy of traditional mitochondrial treatments. Furthermore, the mitochondria demonstrated impressive storage stability, retaining function for 24 hours at 4°C—a critical factor for real-world clinical applications. The concept of organelle tuning, as demonstrated in this study, could potentially be adapted to generate other cellular components, broadening the horizons of cell engineering and therapeutic applications.
“This work represents a paradigm shift in our ability to produce therapeutic mitochondria,” stated corresponding author Dr. Hongwei Ouyang. “By reprogramming stem cells into highly efficient mitochondrial factories, we have solved the critical supply issue that has hindered clinical applications. The mito-condition medium not only amplifies mitochondrial production but also enhances their quality, which directly translates to superior therapeutic outcomes. This platform has the potential to revolutionize treatment strategies for a host of degenerative diseases driven by mitochondrial dysfunction.”
The most immediate application of this technology lies in osteoarthritis treatment, where it offers a promising regenerative solution. However, its impact extends far beyond joint disorders, with potential benefits for conditions such as heart disease, neurodegenerative disorders, and wound healing. By enabling the large-scale production of standardized, high-quality mitochondria, this breakthrough could transition mitochondrial transplantation from an experimental concept to a widely accessible clinical therapy.
Moreover, the organelle-tuning approach introduced in this study may serve as a blueprint for generating other specialized cellular components, unlocking new possibilities in cell-based medicine. While challenges remain in refining delivery mechanisms and assessing long-term effects, this breakthrough represents a significant leap forward in regenerative science, offering renewed hope for millions suffering from mitochondrial-related diseases.
Reference: “Organelle-tuning condition robustly fabricates energetic mitochondria for cartilage regeneration” by Xuri Chen, Yunting Zhou, Wenyu Yao, Chenlu Gao, Zhuomin Sha, Junzhi Yi, Jiasheng Wang, Xindi Liu, Chenjie Dai, Yi Zhang, Zhonglin Wu, Xudong Yao, Jing Zhou, Hua Liu, Yishan Chen and Hongwei Ouyang, 17 March 2025, Bone Research.
DOI: 10.1038/s41413-025-00411-6
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
3 Comments
For me it begs the question: how well will the transplanted mitochondria do in a real world of human diets made toxic with officially (FDA in the US) approved food poisoning (e.g., soy, TBHQ and added MSG, minimally) which tend to acidify blood, consume excessive calcium and deactivate native mitochondria?
Don’t eat that crap anyway. I don’t, and anybody can adjust their diet to eliminate that. Fresh whole foods can be anybody’s priority.
This is a short term study in mice, so long-term impacts are unknown. It is also very cruel and painful to create a model of osteoarthritis in mice, and this is not a good model for human osteoarthritis, which is a long-term degenerative disease. And while some diseases involve mitochondrial dysfunction, the cause of that dysfunction needs to be addressed, instead of just replacing the mitochondria.