Scientists reveal a hidden “wiring system” in brown fat that could help turn your body into a calorie-burning furnace.
Researchers have uncovered how a crucial protein helps activate brown fat by promoting the growth of blood vessels and nerve connections within this heat-producing tissue.
The study, published today (March 25) in Nature Communications, highlights a possible new way to address obesity that focuses on increasing how much energy the body burns rather than simply reducing appetite.
Brown Fat and Its Role in Burning Energy
Most body fat is white fat, which stores excess energy and can contribute to obesity when levels become too high. In contrast, humans and other mammals also have smaller amounts of brown fat, a specialized type of tissue that helps regulate body temperature and is closely tied to metabolism and weight control. When triggered by cold exposure, brown fat uses glucose and lipids to produce heat through a process known as thermogenesis.
“During thermogenesis, all of that chemical energy is dissipated as heat instead of being stored in the body as white fat,” said Farnaz Shamsi, assistant professor of molecular pathobiology at NYU College of Dentistry and the study’s senior author. “By rapidly taking up and using fuel sources from our bodies and the food that we eat, brown fat acts like a metabolic sink that draws in nutrients and prevents them from being stored.”
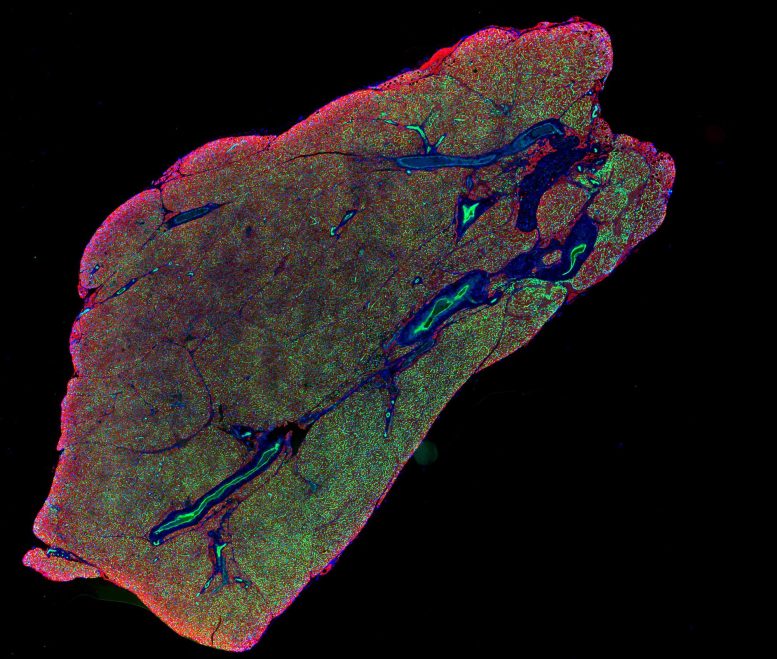
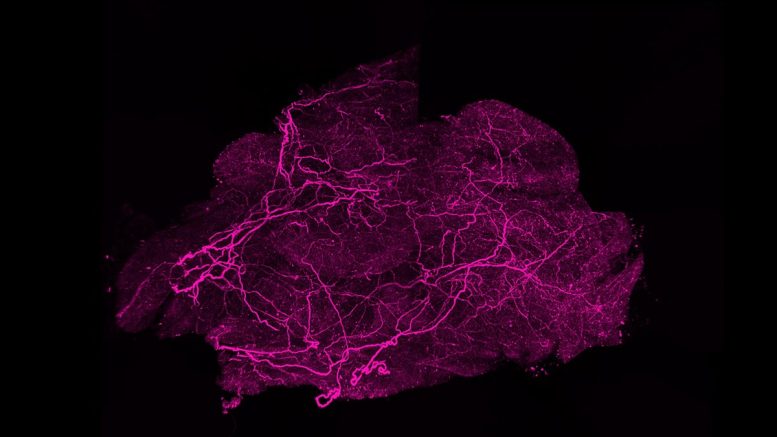
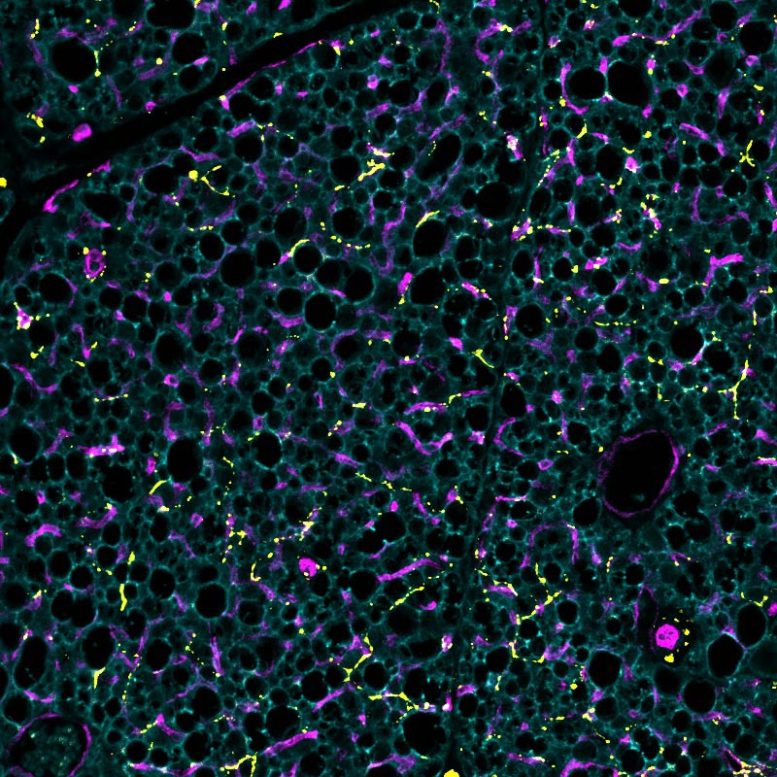
Brown fat depends on dense networks of nerves and blood vessels to function properly. Nerves allow communication with the brain, which signals brown fat to activate when the body senses cold. Blood vessels deliver oxygen and nutrients needed for heat production and help distribute that heat throughout the body. While much research has focused on stimulating fat cells themselves, less attention has been given to how these supporting networks are formed and maintained.
SLIT3 Protein Controls Blood Vessels and Nerves
Earlier work from Shamsi’s lab used single-cell RNA sequencing to identify SLIT3, a protein released by brown fat cells that may help coordinate communication between them. Once produced, SLIT3 is split into two separate fragments.
In the new study, researchers used a range of methods in both human and mouse cells to identify the enzyme BMP1, which cuts SLIT3 into these two parts. They found that each fragment plays a distinct role. One promotes the formation of blood vessels, while the other supports the growth of nerve networks.
“It works as a split signal, which is an elegant evolutionary design in which two components of a single factor independently regulate distinct processes that must be tightly coordinated in space and time,” noted Shamsi.
The team also identified a receptor called PLXNA1 that interacts with one of the SLIT3 fragments to regulate nerve development in brown fat. Experiments in mice showed that removing SLIT3 or the PLXNA1 receptor made the animals more sensitive to cold and less able to maintain body temperature. Further analysis revealed that their brown fat lacked proper nerve structure and sufficient blood vessel density.
Links to Obesity and Metabolic Health
To explore whether these findings apply to humans, researchers analyzed fat tissue samples from more than 1,5000 individuals, including people with obesity. They focused on the gene responsible for producing SLIT3, which earlier studies have linked to obesity and insulin resistance. Their results suggest that SLIT3 gene activity may influence fat tissue health, inflammation, and insulin sensitivity in individuals with obesity.
“That really got our attention, as it suggests that this pathway could be relevant in human obesity and metabolic health,” said Shamsi.
A New Approach to Treating Obesity
Most current weight loss medications, including GLP-1s, work by suppressing appetite and reducing how much people eat. In contrast, targeting brown fat could increase the body’s energy expenditure. The new findings, including how SLIT3 is split and how its fragments interact with receptors to shape nerve and blood vessel networks, point to several possible targets for future therapies.
“Our research shows that just having brown fat isn’t enough—you need the right infrastructure within the tissue for heat production,” said Shamsi.
Reference: “SLIT3 fragments orchestrate neurovascular expansion and thermogenesis in brown adipose tissue” by Tamires Duarte Afonso Serdan, Heidi Cervantes, Benjamin Frank, Akhil Gargey Iragavarapu, Qiyu Tian, Daniel Hope, Chan Hee J. Choi, Anne Hoffmann, Adhideb Ghosh, Christian Wolfrum, Matthew B. Greenblatt, Paul Cohen, Matthias Blüher, Halil Aydin, Gary J. Schwartz and Farnaz Shamsi, 25 March 2026, Nature Communications.
DOI: 10.1038/s41467-026-70310-9
Additional study authors include Tamires Duarte Afonso Serdan, Heidi Cervantes, Benjamin Frank, Akhil Gargey Iragavarapu, Qiyu Tian, Daniel Hope, and Halil Aydin of NYU College of Dentistry; Chan Hee Choi and Paul Cohen of Rockefeller University; Anne Hoffmann and Matthias Blüher of the University of Leipzig; Adhideb Ghosh and Christian Wolfrum of ETH Zurich; Matthew Greenblatt of Weill Cornell Medical College; and Gary Schwartz of Albert Einstein College of Medicine.
The research was supported in part by the National Institutes of Health (K01DK125608, R03DK135786, R01DK136724, RC2DK129961, R35GM150942), the G. Harold and Leila Y. Mathers Charitable Foundation, the American Heart Association (24CDA1271852), the Einstein-Mount Sinai Diabetes Center, the NYU Dentistry Department of Molecular Pathobiology, and the Boettcher Foundation.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
11 Comments
I’ve been waiting for the magic pill my entire life already! Hurrrrry!
How do we keep check on which companies start researching this with intentions to develop a usable product ?
I want to be a participant in that Research Trial.
Capsiniods (synthetic or naturally extracted from peppers) can activate and promote calorie burning brown fat but appetite suppression would likely still be needed for most obese people to lose weight, in fact the body burning extra calories usually makes people hungry.
How can I become a participant in this study ?
Low carb high protein intake is the way to go. East a little carb close to exercise before or after and aim for 2 grams per 1lb of desired body weight. Slow walking for around 15 mins after every meal.
I wabt to be part of this trail
I’d like to know more about the research and possibly participate in the trial.
Low carb high protein intake is the way to go. East a little carb close to exercise before or after and aim for 2 grams per 1lb of desired body weight. Slow walking for around 15 mins after every meal.
2 grams of protein per 1lb of desired bodyweight.
A good bout of gastro-enteritis and the weight just drops off.