Scientists found that carbon enabled Earth’s core to solidify. Without it, the inner core may not exist.
A team of scientists from the University of Oxford, University of Leeds, and University College London has uncovered new evidence about the chemistry of Earth’s core by identifying how it was able to crystallize millions of years ago. Their findings were recently published in Nature Communications.
The study revealed that Earth’s core would need to contain about 3.8% carbon for crystallization to begin. This suggests that carbon may be more abundant in the core than previously believed, and that it played an essential role in enabling the freezing process, offering a rare view into the hidden dynamics deep within the planet.
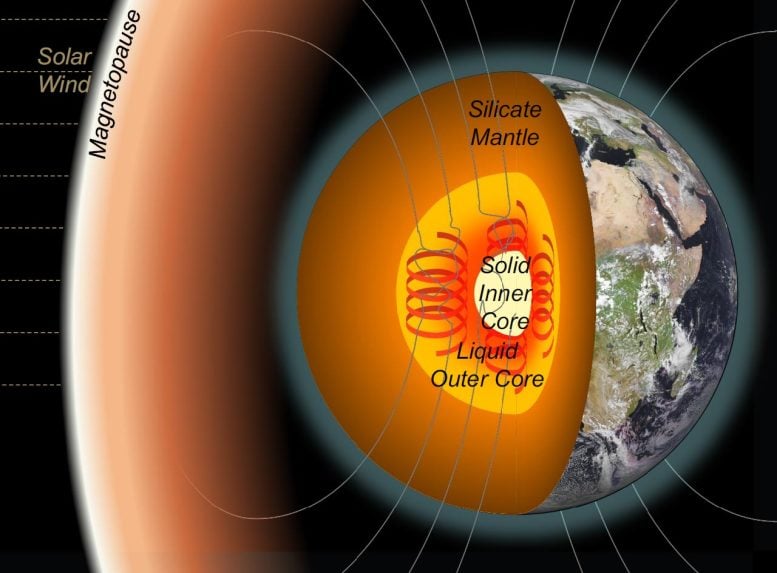
At the planet’s center lies the inner core, a dense, iron-rich solid mass that continues to grow as the surrounding molten outer core gradually cools and solidifies. Exactly how this process began, however, has been the subject of long-standing scientific debate.
The challenge of supercooling
The formation of the inner core is not simply a matter of the core reaching its freezing temperature. Instead, crystallization depends heavily on its chemical makeup. Molten iron, like water droplets in clouds, must undergo supercooling (cooling below its normal melting point) before solidification can occur.
Earlier models estimated that 800 to 1000 °C of supercooling would be required for a pure iron core to freeze.
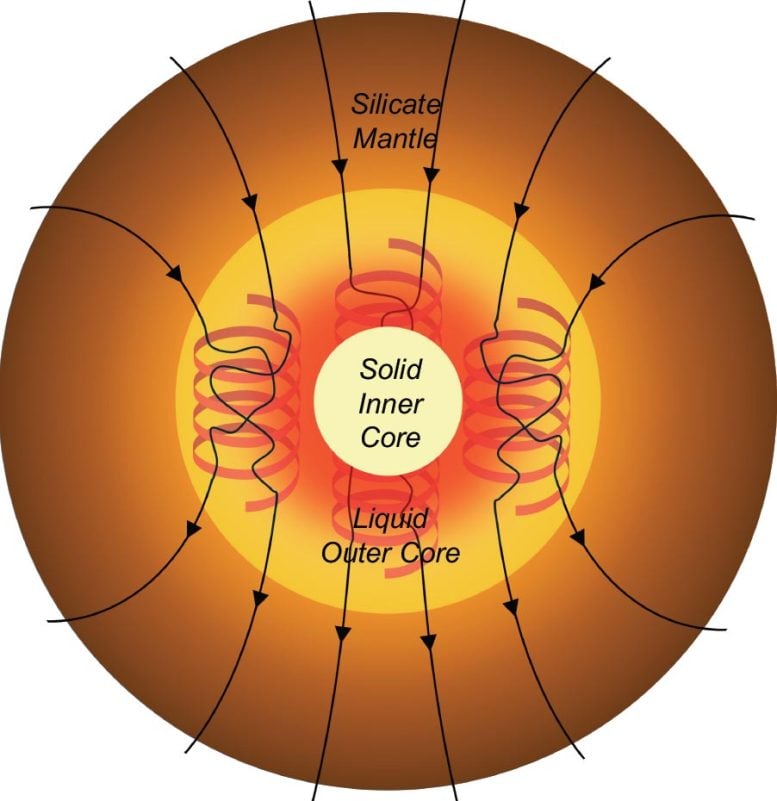
If the Earth’s core had cooled by that much, models indicate the inner core would have grown to an unrealistic size and the planet’s magnetic field would have collapsed. Neither outcome matches Earth’s history, so scientists have proposed that, at most, the core cooled only about 250 °C below its melting point.
Simulating the deep Earth
This new research aimed to understand how the inner core exists as observed today, with such limited supercooling in the past. Without direct access to the Earth’s deep interior, the research team needed to rely on computer simulations of the freezing process.
They looked at the presence of other elements, specifically silicon, sulfur, oxygen, and carbon, and how these might affect the freezing process.
“Each of these elements exists in the overlying mantle and could therefore have been dissolved into the core during Earth’s history,” explained co-author Associate Professor Andrew Walker (Department of Earth Sciences, University of Oxford). “As a result, these could explain why we have a solid inner core with relatively little supercooling at this depth. The presence of one or more of these elements could also rationalize why the core is less dense than pure iron, a key observation from seismology.”
Testing elements in simulations
Using atomic-scale computer simulations of around 100,000 atoms at supercooled temperatures and pressures equivalent to those in the inner core, the research team tracked how often small crystal-like clusters of atoms formed from a liquid. These “nucleation” events are the first steps toward freezing.

What they found was surprising: silicon and sulfur, elements often assumed to be present in the core, actually slow down the freezing process. In other words, more supercooling would be needed to start forming the inner core if these elements were abundant in that part of the Earth.
On the other hand, they found that carbon helped to accelerate freezing in the simulation.
Carbon as the key factor
In the study, the researchers tested how much supercooling would be required to freeze the inner core if 2.4% of the core’s mass were made of carbon. The result: about 420 °C, still too high, but the closest result to viability yet.
But when they extrapolated their results to a case where 3.8% of the core’s mass is carbon, the required supercooling dropped to 266 °C. This is the only known composition that could explain both the nucleation and the observed size of the inner core.
This result indicates that carbon may be more abundant in Earth’s core than previously thought, and that without this element, the formation of a solid inner core may never have happened.
Freezing without nucleation seeds
The experiments also show that inner core freezing was possible with just the right chemistry, and unlike water when it forms hail, it did so without “nucleation seeds,” tiny particles which help to initiate freezing. This is vital, because when tested in previous simulations, all of the candidates for nucleation seeds in the core have melted or dissolved.
Lead author Dr. Alfred Wilson (School of Earth and Environment, University of Leeds) said: “It is exciting to see how atomic-scale processes control the fundamental structure and dynamics of our planet. By studying how Earth’s inner core formed, we are not just learning about our planet’s past. We’re getting a rare glimpse into the chemistry of a region we can never hope to reach directly and learning about how it could change in the future.”
Scientists have debated when the inner core began to solidify for decades, with some arguing for an ancient inner core (with freezing beginning more than two billion years ago) and others suggesting a much younger age (less than half a billion years). With this new information about the carbon content of the core, we are one step closer to constraining its chemistry and physical properties, and therefore how it evolved.
Reference: “Constraining Earth’s core composition from inner core nucleation” by Alfred J. Wilson, Christopher J. Davies, Andrew M. Walker and Dario Alfè, 4 September 2025, Nature Communications.
DOI: 10.1038/s41467-025-62841-4
The work was funded by the Natural Environment Research Council (NERC).
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
So the Earth is a Tootsie Pop?