
Photosynthesis is a vital process enabling plants to transform carbon dioxide into organic compounds using sunlight. The Light-harvesting complex II (LHCII) consists of pigment molecules attached to proteins. It alternates between two primary roles: when under intense light, it dissipates excess energy as heat through nonphotochemical quenching, and under low light, it efficiently transfers light to the reaction center.
Recent bioengineering research has revealed that speeding up the switch between these functions can boost photosynthetic efficiency. For instance, soybean crops have shown yield increases of up to 33%. However, the precise atomic-level structural changes in LHCII that trigger this regulation were previously unknown.
Innovative Research Approach
ln a new study, researchers led by Prof. Weng Yuxiang from the Institute of Physics of the Chinese Academy of Sciences, together with Prof. Gao Jiali’s group from Shenzhen Bay Laboratory, combined single-particle cryo-electron microscopy (cryo-EM) studies of dynamic structures of LHCII at atomic resolution with multistate density functional theory (MSDFT) calculations of energy transfer between photosynthetic pigment molecules to identify the photosynthetic pigment quantum switch for intermolecular energy transfer.
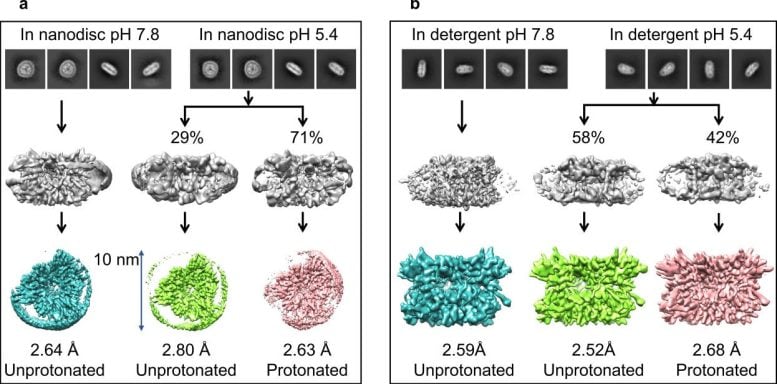
As part of their work, they reported a series of six cryo-EM structures, including the energy transfer state with LHCII in solution and the energy quenching state with laterally confined LHCII in membrane nanodiscs under both neutral and acidic conditions.
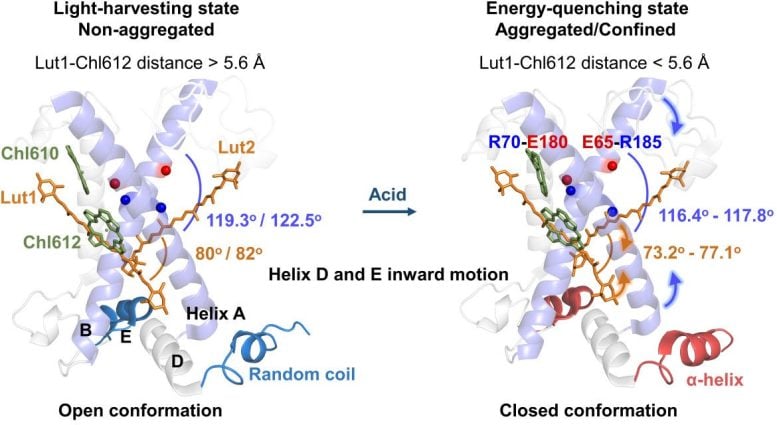
Comparison of these different structures shows that LHCII undergoes a conformational change upon acidification. This change allosterically alters the inter-pigment distance of the fluorescence quenching locus Lutein1 (Lut1)–Chlorophyll612 (Chl612) only when LHCII is confined in membrane nanodiscs, leading to the quenching of excited Chl612 by Lut1. Thus, LHCII confined with lateral pressure (e.g., aggregated LHCII) is a prerequisite for non-photochemical quenching (NPQ), whereas acid-induced conformational change enhances fluorescence quenching.
Quantum Switching Mechanism in Photosynthesis
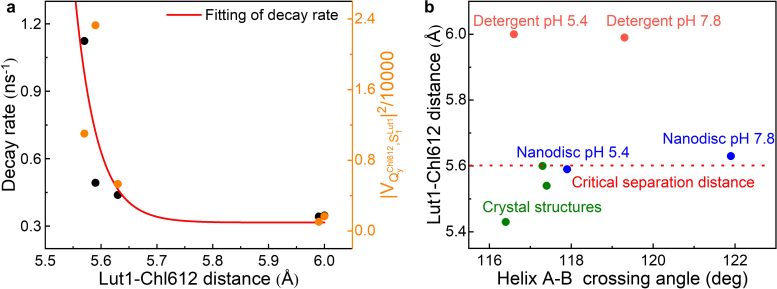
Through MSDFT calculations of cryo-EM structures and the known crystal structure in quenched states, together with transient fluorescence experiments, a significant quantum switching mechanism of LHCII has been revealed with Lut1–Chl612 distance as the key factor.
This distance regulates the energy transfer quantum channel in response to the lateral pressure on LHCII and the conformational change, that is, a slight change at its critical distance of 5.6 Å would allow reversible switching between light harvesting and excess energy dissipation. This mechanism enables a rapid response to changes in light intensity, ensuring both high efficiency in photosynthesis and balanced photoprotection with LHCII as a quantum switch.
Previously, these two research groups had collaborated on molecular dynamics simulations and ultrafast infrared spectroscopy experiments and had proposed that LHCII is an allosterically regulated molecular machine. Their current experimental cryo-EM structures confirm the previously theoretically predicted structural changes in LHCII.
Reference: “Cryo-EM structures of LHCII in photo-active and photo-protecting states reveal allosteric regulation of light harvesting and excess energy dissipation” by Meixia Ruan, Hao Li, Ying Zhang, Ruoqi Zhao, Jun Zhang, Yingjie Wang, Jiali Gao, Zhuan Wang, Yumei Wang, Dapeng Sun, Wei Ding and Yuxiang Weng, 31 August 2023, Nature Plants.
DOI: 10.1038/s41477-023-01500-2
This research was supported by projects from the Chinese Academy of Sciences, the National Natural Science Foundation of China, and the Shenzhen Municipal Science and Technology Innovation Commission.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.