Researchers have uncovered an unexpected biological process that may allow the body to directly control stored sugar, challenging decades of scientific understanding.
Researchers at WEHI have identified a previously unknown way the body controls sugar storage, a discovery that challenges long-standing biology concepts and could open new directions for disease treatment.
Published in Nature, the study describes a potential method for directly reducing glycogen, the stored form of sugar in the body.
The findings could eventually help people with conditions linked to excess sugar buildup, including diabetes, heart disease, and several rare disorders that currently lack effective treatments.
At a glance
- WEHI scientists discovered that glycogen can be directly regulated by ubiquitin, a protein best known for marking damaged proteins for recycling or removal.
- The study is the first to show that ubiquitin can regulate glycogen in humans, overturning more than 50 years of scientific understanding.
- Using a newly developed technique, the team uncovered a pathway that could lead to future treatments for diseases caused by abnormal glycogen storage, including liver disease, heart disease, and rare Glycogen Storage Diseases.
How Glycogen Works in the Body
After consuming sugar, the body converts excess amounts into glycogen, which is mainly stored in the liver and muscles.
Scientists have studied glycogen metabolism for generations, and the process has long been considered fully understood.
Professor David Komander, co-lead author of the study and head of WEHI’s Ubiquitin Signaling Division, said the findings fundamentally expand current knowledge of biology.
“It’s quite likely biology books will need to be amended as a result of our findings,” Prof. Komander, also head of WEHI’s Ubiquitin Signaling Division, said. “We’ve uncovered a second pathway where glycogen can be directly regulated—likely on demand. This is an exciting breakthrough for people living with diseases caused by excessive glycogen.”
Excess Glycogen Linked to Rare and Common Diseases
Glycogen Storage Diseases (GSD) are rare inherited conditions that prevent the body from properly producing or breaking down glycogen. Many of these disorders still have no treatment options.
Excess glycogen is also associated with more common health problems, including diabetes, obesity, liver disease, and heart disease.
These conditions are driven by glycogen buildup, yet no current therapies can directly target the glycogen molecule itself.

Why Targeting Glycogen Matters
“Exciting new drugs—such as Ozempic—are transforming how we manage blood sugar, indirectly via hormonal regulation,” Prof. Komander said.
“Without being able to regulate glycogen itself, it is hard to combat its accumulation—the root cause of many diseases. That’s why our study is exciting. We’ve found a way to go straight to the source.”
Ubiquitin is widely known for attaching to proteins as part of the body’s system for identifying and removing damaged material.
Glycogen, however, is a sugar rather than a protein. Despite this, the researchers discovered that ubiquitin can also attach to sugars in both animal models and human cells.
Ubiquitin’s Surprising Role
Co-lead author Dr. Simon Cobbold said the discovery highlights an overlooked role for ubiquitin.
“Ubiquitin is really an unsung hero that has been quietly working in the background all this time, keeping us alive,” Dr. Cobbold said.
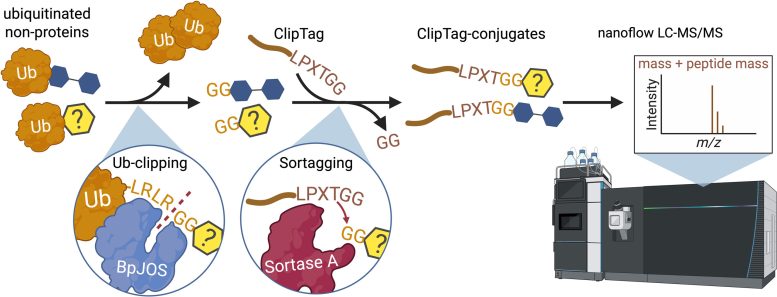
The breakthrough relied on NoPro-clipping, an advanced technique developed over four years by Dr. Cobbold, Prof. Komander, and the first author, Marco Jochem.
The method allows researchers to study ubiquitin in far greater detail and, for the first time, detect non-protein ubiquitination events using mass spectrometry.
NoPro-Clipping Opens New Possibilities
“Without our tools and method, this remarkable process would have remained invisible,” Dr. Cobbold said. “That’s the beauty of NoPro-clipping—it’s allowing us to study a canvas of molecules the ubiquitin field has overlooked all this time.”
PhD student Marco Jochem said the technology has broad potential applications.
“Not only can we use it to detect ubiquitinated glycogen—we can also uncover ubiquitinated metabolites like glycerol and spermine, which we’ve discovered for the first time in all our cells,” he said. “Our discovery is rewriting the fundamental rules of biology and ubiquitin signaling. And I’m sure we’ve only hit the tip of the iceberg.”
Ubiquitin Tags Surge as Glycogen Breaks Down
In one of the study’s most important findings, researchers used NoPro-clipping to visualize how ubiquitin attaches to glycogen in the livers of mice during feeding and fasting.
When mice entered a fasting state and required energy, glycogen levels dropped.
At the same time, ubiquitin “tags” increased as glycogen was depleted, suggesting that sugar ubiquitination helps regulate glycogen breakdown.
The results identify ubiquitin as a previously unknown component of glycogen metabolism and add a new layer to a biochemical process long considered settled science.
A Potential New Strategy to Fight Sugar-Driven Diseases
The researchers found that changing ubiquitin tags on sugar appears to influence how and when glycogen is released.
They also showed that increasing glycogen ubiquitination reduced glycogen levels inside cells.
If the results can eventually be replicated in animals and humans, the discovery could lead to entirely new approaches for treating disease. Early discussions with investors are already underway.
Reference: “Ubiquitination of glycogen and metabolites in cells and tissues” by Marco Jochem, Simon A. Cobbold, Craig A. Goodman, Catharina Kueng, Anthony Cerra, Laura F. Fielden, Man Lyang Kim, Philipp Schenk, Ria Agarwal, Xiangyi S. Wang, Simon R. Scutts, Michael Pandos, Lin Tang, Thomas Hermanns, Shane M. Devine, Martin Brzozowski, Yuri Shibata, Niall D. Geoghegan, Catriona A. McLean, Bernhard C. Lechtenberg, Kay Hofmann, Paul Gregorevic and David Komander, 22 April 2026, Nature.
DOI: 10.1038/s41586-026-10548-x
The research is supported by the National Health and Medical Research Council (NHMRC), National Institutes of Health (NIH), and Victorian Government.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
1 Comment
So this would only help those whose Glycogen Storage Diseases (GSD) cause an excess of glycogen to be stored but not those who have trouble storing glycogen in the first place? Or would this suggest those folks who have trouble storing glycogen already have high levels of ubiquitin?